Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 15.01.2026
ResMed AirSense 10 AutoSet CPAP Device Analysis Report
Device Identification
The product analyzed in the visuals is a positive airway pressure (PAP) device used in the treatment of sleep apnea. The product consists of a main control unit, an integrated humidifier water tank, and an original carrying bag. The product falls into the category of a home medical device.
Brand and Model
As a result of detailed visual inspections, the brand and model of the device have been identified unequivocally. In the second visual, the text and logo located on the front face of the device, at the bottom right of the screen, are clearly legible.
- Brand: ResMed
- Model: AirSense 10 AutoSet
Authenticity Assessment
The product’s general material quality, plastic texture, user interface font on the screen, and menu design are fully consistent with ResMed’s standard design language. Furthermore, the multilingual warning labels (humidifier emptying warning) found inside the carrying bag shown in the third visual, along with the bag’s stitching/fabric structure, indicate that the product and its accessories are original.
Areas of Use
This device is designed for the treatment of patients with Obstructive Sleep Apnea (OSA). Thanks to its “AutoSet” feature, the device automatically adjusts pressure according to the user’s breathing, ensuring the airway remains open. It is suitable for use in hospital settings or for individual treatment at home. With its integrated humidifier, it provides comfort by humidifying the air the patient breathes.
Quantity Information
The number of physical parts identified in the visuals is as follows:
- 1 unit of ResMed AirSense 10 main unit device.
- 1 unit of transparent water tank integrated into the device (HumidAir).
- 1 unit of original grey carrying bag.
General Condition and State
The device generally has a clean and well-maintained appearance. No significant heavy damage marks are visible on the body. In the first visual, the device appears to be operational and its screen light is bright, which proves its electronic activity. The device has a usage period that can be described as “lightly used”.
Physical Deformation Examination
No deep scratches, dents, or dings have been detected on the outer casing of the device. The black textured surface (first visual) appears clean. However, in the second visual, there are superficial dustings on the device’s shiny surfaces (especially the top part and around the button) resulting from normal use or environmental factors. The water tank is transparent and, as far as can be seen from the outside, shows no signs of calcification or yellowing.
Mechanical and Electronic Components
Mechanical: The hinge of the cover on the right side of the device (water tank inlet), the start/stop button on the top, and the round control dial on the side appear visually sound.
Electronic: In the first visual, the screen is seen working flawlessly, with no dead pixels or stains, and the colors and texts are clear. Furthermore, the white backlight under the “Ev” (Home) button has been detected as active. The device’s software is operating stably.
Accessories and Carrying Bag
In the visuals, the device is shown inside its original grey carrying bag (with ResMed logo). The blue Velcro fastening straps inside the bag appear sturdy. As indicated by the icons inside the bag, special compartments for adapters and cables are present; however, the power cable, mask, and air hose are not explicitly displayed in the visuals, only their placement icons are visible.
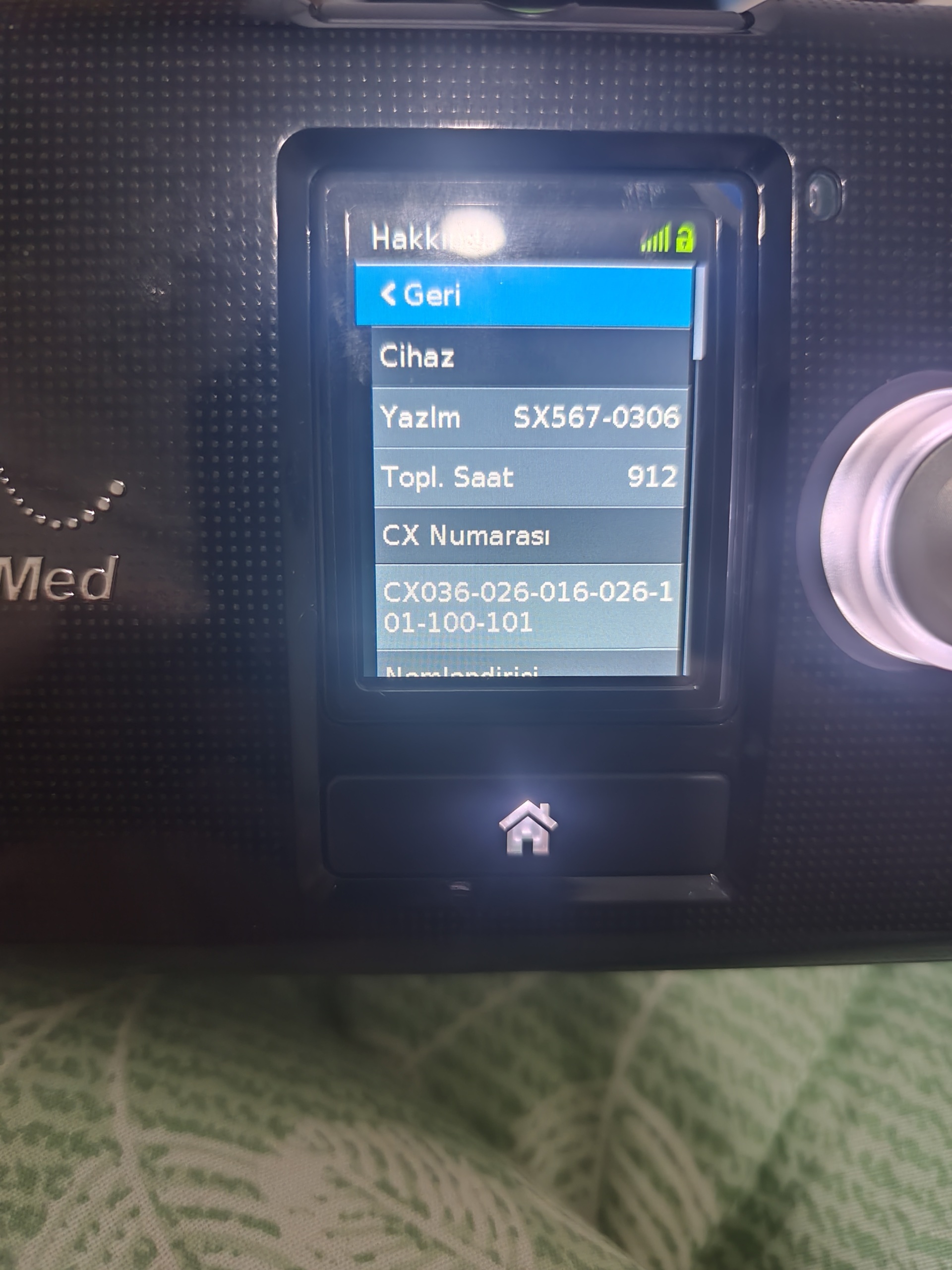
Screen Analysis and Usage Duration
The device’s “Hakkında” (About) menu is clearly displayed. The data obtained from this screen provides the most accurate information regarding the device’s usage history:
- Topl. Saat (Total Hours): 912 Hours
- This data indicates that the device has been used but is still very early in its lifespan for a respiratory device (These devices can generally be used for up to 15,000-20,000 hours).
Labels and Technical Information
The technical data visible on the screen interface is as follows:
- Software Version: SX567-0306
- CX Number: CX036-026-016-026-1 01-100-101
Furthermore, the label on the bag contains warnings in English, German, and French regarding the humidifier needing to be emptied before transporting the device (Ensure humidifier is EMPTY / Stellen Sie vor dem Transport sicher / Veillez à ce que le réservoir).
Battery Status
The device is shown in the visual operating while connected to electricity. These models typically operate with mains electricity. No battery unit, either attached to the device or external, is visible in the visuals.
Current Faults and Risk Assessment
There are no error messages or physical breakages in the visuals indicating that the device is faulty. No warning icon is present on the screen.
Potential Risk: As the device contains electronic components, it is sensitive to water contact. While no risk is observed in the visual, ensuring the water tank is empty during transport (as also stated on the bag cover) is the most important rule of use.