Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 23.09.2025
RespiroX Auto CPAP Device Analysis Report
Overview and Product Description
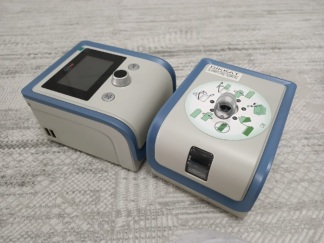
The image shows an Auto CPAP device with the brand name RespiroX. This device is an automatic pressure-adjusting respiratory support unit used in the treatment of breathing disorders such as obstructive sleep apnea (OSA). It is understood that the device has an integrated humidifier unit designed to increase user comfort. The product is located inside a black, zippered carrying case, which is presumed to belong to it.
Box Contents and Quantity Information
The products and their quantities shown in the visuals are listed below:
- 1 RespiroX Auto CPAP device
- 1 device-specific carrying case
Other essential accessories required for the device’s operation, such as a power cable, respiratory hose, or mask, are not present in the visuals.
Condition and State Assessment
The device generally appears lightly used and in very good condition. It is observed to be physically clean and carefully maintained. The fact that the device is stored inside its carrying case indicates it has been well-protected. There are no signs of wear or use-related stains on its surface.
Physical and Mechanical Analysis
No deformation such as scratches, cracks, dents, or color fading has been detected on the product’s casing, screen, or control panel. The buttons on the control panel and their symbols are unworn and clearly legible. Mechanical parts such as the humidifier chamber and filter cover located at the bottom of the device are in place and visually free of any damage. The zippers of the carrying case also appear functional and robust.
Electronic and Screen Analysis
The device’s digital screen is off, therefore no operational data, menu content, or error messages can be displayed. No scratches or defects have been detected on the physical surface of the screen. The control panel contains electronic control buttons such as the power button, humidifier level, and pressure adjustment buttons.
Label and Information Texts
An important warning label regarding the use of the humidifier unit is located on the device. This label contains the following text:
- “CAUTION !!! DO NOT CARRY WITH WATER IN THE HUMIDIFIER UNIT, OTHERWISE YOUR DEVICE WILL BE OUT OF WARRANTY”
This warning states that the device should not be carried with water in the humidifier unit, and failure to comply with this will void the product’s warranty. No other labels containing technical details such as serial number, lot number, REF code, or production date are visible in the visuals.
Potential Malfunction Risk
During the examination based on the visuals, no concrete findings suggesting that the device might malfunction in the near future have been encountered. The product’s overall condition is very good, and there are no visible signs of wear, damage, or neglect. This situation is a positive indicator, strengthening the likelihood that the mechanical and electronic components of the device are in perfect working order.