Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 07.10.2025
Vestel Medicraft Bilevel Device Analysis Report
Overview and Device Identification
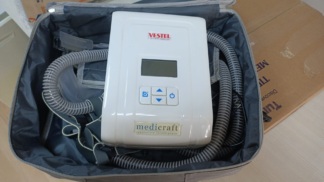
The product shown in the images is a Bilevel device used for respiratory therapy. The word “BILEVEL” is clearly visible on the product. Such devices are medical equipment that provide positive airway pressure, commonly used in the treatment of respiratory conditions like sleep apnea. The image only shows the device itself, and there is one unit in total.
Brand and Model Information
A detailed examination revealed three different brand logos and names on the device. These are Vestel, Medicraft, and Bilevel, respectively. It is understood that the device is a “Bilevel” series respiratory device produced by Medicraft and marketed through a partnership or distribution by Vestel. The brand and model inscriptions are clear and appear to be original prints. This information leaves a positive impression regarding the product’s authenticity.
Physical Condition and Deformation Assessment
The overall condition of the device is assessed as used. There are some noticeable physical deformations.
- Casing: The beige plastic casing of the device has small scratches and slight scuff marks in various places, particularly on the corners and edges.
- Dirt and Discoloration: There is general dirtiness on the casing and small stains noticeable in some areas. However, no significant color fading or yellowing that would be bothersome has been observed.
- Integrity: No cracks, breaks, or major impact marks were found. The device maintains its single-piece integrity in terms of its casing.
No abnormalities were detected in the visible mechanical components such as the button array and casing joints. All buttons are in place and appear physically sound.
Electronic Components and Accessories
Limited information about the device’s electronic components can be gathered from the images. The front panel features one digital LCD screen and four control buttons. Since the device is not operational, no comments can be made regarding the screen’s functions, but there are no cracks or physical damage to the screen. The images do not show a power cable, respiratory mask, hose, or any other accessories belonging to the device. As the rear or side faces of the device are not visible, the socket inputs and connection ports could not be analyzed. There is also no visual evidence regarding the presence or condition of its battery.
Label and Technical Information
No labels or information plates containing the device’s serial number, REF code, manufacturing year, or other technical specifications are visible in the images. Therefore, this information cannot be included in the report.
Risk Assessment
Based on the examination of the images, there is no serious damage (such as cracks, breaks, etc.) to the device’s casing. This suggests that the device may have been used carefully. However, superficial scratches and dirt resulting from its used condition are present. The greatest potential risk of the device is the absence of any accessories and the inability to test it in working condition. Without cables and other accessories, a clear comment cannot be made about the device’s functional status. Nevertheless, the absence of visibly severe wear or damage suggests that the device’s overall condition is good.