Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 16.02.2026
Philips Respironics DreamStation Respiratory Device Analysis Report
Device Identification
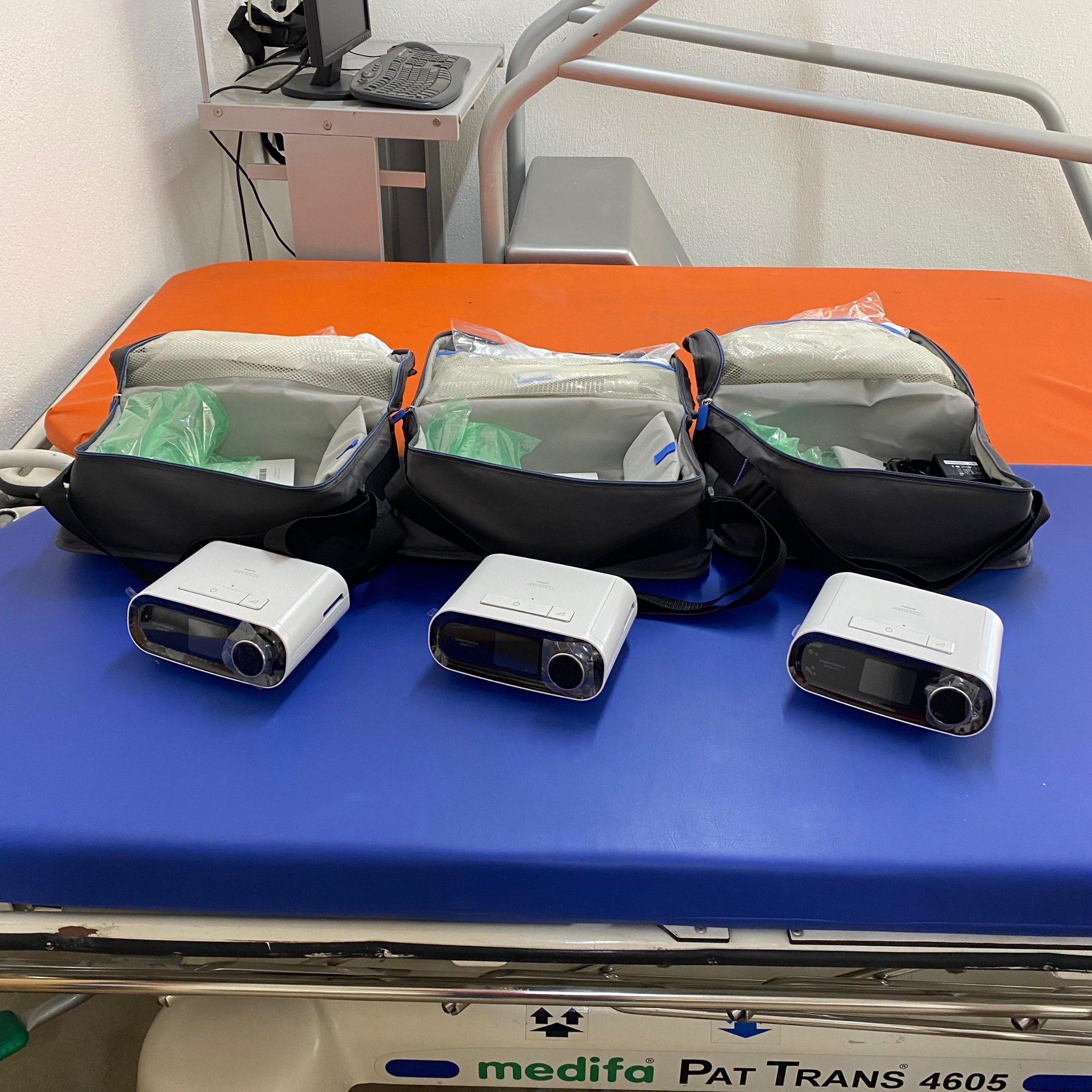
As a result of the analysis performed on the visuals, three respiratory therapy devices (CPAP/BiPAP) and their carrying bags were identified at the main focal point. The devices are suitable for desktop use, compact in structure, and can be used by healthcare professionals or home care patients.
Brand and Model
Upon examining the physical structure of the devices, button layout, screen position, the circular adjustment knob located on the right side, and the design details on the front panel, it is clearly understood that the products belong to the Philips Respironics brand. Considering the text format indicating the model series on their front panels and the iconic design of the device, it has been confirmed that the products are from the DreamStation series (likely DreamStation CPAP or BiPAP models).
Authenticity
The molding craftsmanship, material texture, color harmony, and the placement of logos (brand and model on the front panel) of the devices perfectly match manufacturer standards. The original carrying bags next to them and the packaged accessories inside also support their authenticity. Based on visual evidence, the products are evaluated as original.
Areas of Use
These devices are used to provide positive airway pressure to keep the airway open in the treatment of sleep apnea (OSAS) and in cases of respiratory failure. They are suitable for use in home care settings, sleep laboratories, or pulmonary departments of hospitals.
Quantity Information
The visual shows a total of 3 main units (devices) and 3 original carrying bags standing behind each device. The total number of sets is 3.
General Condition
The devices appear cosmetically clean and in good condition. There is no noticeable yellowing or heavy dirt on the white color of their outer casings. The overall condition of the products gives the impression of “lightly used” or “clean second-hand.” The open posture of the bags and the arrangement of the materials inside indicate that the devices have been stored and carefully kept.
Physical Deformation
During the inspection, no cracks, deep scratches, or dents were detected on the visible front panels, top covers, or side surfaces of the devices. The integrity of the plastic components has been preserved. No impact marks were found on the screen surfaces.
Mechanical Components
The control knobs (circular buttons) located on the right side of the devices are in place and appear physically sound. No visually apparent malfunction or missing part has been observed in the on/off mechanisms or filter covers on the top lid of the devices.
Electronic Components
In the visuals, the devices are not plugged in or operating; therefore, the screens are off (black screen). Whether their electronic functions are working cannot be confirmed visually. However, the physical integrity of the screens is complete; no signs of damage such as liquid contact or screen breakage are visible.
Accessories
- Carrying Bags: A total of 3 original gray carrying bags are present, one for each device. The bags appear sturdy and their zippers are functional (open).
- Content Packages: Inside the bags, there are sealed packages wrapped in green pouches, possibly belonging to tubing or mask components.
- Power Adapters: A black power adapter and its cable can be distinguished inside the rightmost bag. It is highly probable that similar accessories are present in the other bags, but the one clearly visible is the rightmost bag.
- Documents: White booklets/manuals are visible in the pocket sections of the bags.
Battery Status
These models of devices typically operate with an external power adapter and do not standardly contain an internal battery. No integrated battery pack is visible in the visual either.
Label Information
Since the visuals show the front and top angles of the devices, it is not possible to read the technical label (Serial Number, REF code, Year of Manufacture, etc.), which is usually located on the bottom of the device.
Screen Analysis
The color LCD screens located on the front left side of the devices are in the off position. No error codes, warning messages, or menu displays are present on the screen.
Size and Compatibility
The devices are standard-sized (approximately the size of a bedside clock) home-use models designed for adult patients. They fit perfectly into their original carrying bags.
Potential Malfunction Risk
No risk factors that could endanger the physical condition of the devices (e.g., rusting, broken cables, melting of the casing) have been observed in the visuals. The condition of the devices is reassuring and they do not show any signs of risk in terms of their external appearance.