Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 15.11.2025
ResMed S9 VPAP ST Respiratory Device Analysis Report
Overview and Product Description
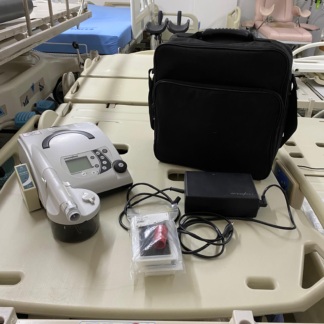
The images show a VPAP (Variable Positive Airway Pressure) device and its accessories used for respiratory therapy. This set consists of a main unit, a humidifier, a power cable, user manuals, and a carrying bag. The products are presented as a complete set, with a total of 7 pieces. The device is designed to operate in Spontaneous/Timed modes to support the patient’s breathing or to trigger respiration at a specific rate in cases of insufficient breathing.
- Main Device Quantity: 1
- Humidifier Unit Quantity: 1
- Power Cable and Adapter Quantity: 1
- Carrying Bag Quantity: 1
- User Documents Quantity: 3
Brand and Model Information
As a result of detailed visual analyses, the brand and model of the device have been clearly identified. The ResMed brand is clearly visible on the front face of the main device, on the carrying bag, and in the user manuals. The model of the device is understood to be S9 VPAP ST, as indicated by the text next to the device’s screen and in the relevant manuals. This model is part of ResMed’s S9 series product family.
Physical Condition and Deformation Analysis
The product’s overall condition appears good and lightly used. No significant scratches, dents, cracks, or discoloration have been observed on the device’s silver-colored casing and black plastic components. The humidifier unit and water chamber are clean and spotless. The carrying bag also has a clean and unblemished appearance. There is a small, white label on the device, presumably indicating it belongs to a patient.
Mechanical and Electronic Components Examination
The device’s mechanical and electronic components are visually in good condition. The control dial and buttons are physically intact. The humidifier unit’s lid mechanism appears sturdy, and the water chamber fits securely in place. The water level indicators inside the chamber are clearly visible. The device’s screen is off but is physically intact and scratch-free. There are no signs of crushing or wear on the power cable and adapter.
Accessories and Equipment
The set includes the main accessories required for the device’s basic use. A respiratory hose (patient circuit) and mask are not present in the images.
- Carrying Bag: The original black carrying bag with the ResMed logo is present and in good condition.
- Power Adapter and Cable: The power unit required for the device’s operation is included in the set.
- Humidifier Unit: The heated humidifier, understood to be model H5i, and its transparent water chamber are included in the set. The unit appears clean and ready for use.
- User Documents: Two “Welcome Guides” prepared for the S9 Series and one “S9 SD Card” informational brochure are present.
Label and Document Analysis
Labels containing the device’s serial number, REF code, or manufacturing date are not clearly legible in the images. On the white label on the device, the beginning of a handwritten text, “TO THE PATIENT…”, can be discerned. The available documents are user manuals only, not warranty certificates or invoices. Therefore, no comment can be made regarding the device’s warranty status or service life.
Potential Risk Assessment
Based on visual inspections, no critical conditions (broken parts, rust, severe crushing of cables, etc.) that would pose an immediate risk of failure have been identified on the device. The product’s generally well-maintained and clean condition suggests that it has been properly stored. In this condition, the device is expected to have reliable operational potential. The missing respiratory hose and mask need to be procured.