Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 28.03.2026
Report code: 1774687812
Respiro Respiratory Device Analysis Report
Device Identification and Areas of Use
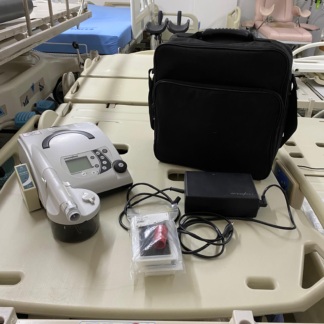
The product shown in the images is a home-use respiratory support device used in the treatment of sleep apnea and respiratory failure. The device is designed to provide positive airway pressure to the patient. It is suitable for use in home or clinical settings, within the fields of pulmonology and sleep medicine.
Brand and Authenticity Assessment
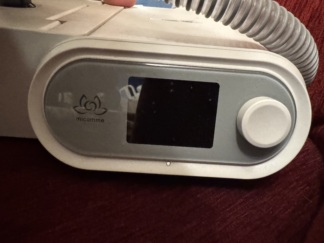
The Respiro brand is clearly visible on the upper part of the screen on the device’s front panel. No label or indication specifying the device’s model is present in the images. Upon examining the device’s casing craftsmanship, the structure of the port entries, and the overall material quality, the product is assessed to be original.
General Condition and Physical State
The device’s general condition is very good. The presence of the factory-applied protective film with a green pull tab on the screen indicates that the device is new or has been used very little. No scratches, dents, cracks, dirt, or discoloration have been detected on the device’s plastic outer casing.
Mechanical and Electronic Components
There is no physical deformation on the central rotary control knob and the two push buttons (alarm mute and ramp/start) located on the front panel. The air outlet port located at the rear and the filter cover in the lower right corner are in good condition. No visible damage or oxidation is present on the electronic connection points.
Connection Points and Interface
The connection points located on the device’s rear panel have been examined in detail, and the following slots have been identified
- One power adapter input socket
- One standard USB port
- One SD card slot (A blue SD card is inserted into the slot)
Accessories and Quantity Information
A total of 1 main respiratory device is present in the images. The accessories provided with the device and located inside the bag are as follows
- One black, zippered, original carrying bag with internal compartments
- One external power adapter (black block-shaped) and its associated power cables
Additional parts necessary for the device’s use, such as a breathing tube, mask, or water chamber, are not present in the images.
Screen Analysis
The device’s LCD screen is in the off position. Therefore, no menu, warning, error message, or usage time information is visible on the screen. There is no physical damage to the screen panel.
Missing Information
Since the technical labels on the bottom or side parts of the device are not visible in the images, no data regarding the serial number, lot number, REF code, manufacturing year, and battery status could be obtained. Furthermore, documents such as an invoice or warranty certificate are not present in the images.
Potential Malfunction Risk
No broken parts, crushed cables, casing wear, or mechanical defects that would prevent the device from operating have been observed in the images. The product’s physical condition is excellent, and based on the visual data, the device is assessed to be in good condition, posing no significant potential malfunction risk.