Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 24.09.2025
Sleptime Plus CPAP/BPAP Device Analysis Report
Overview and Device Description
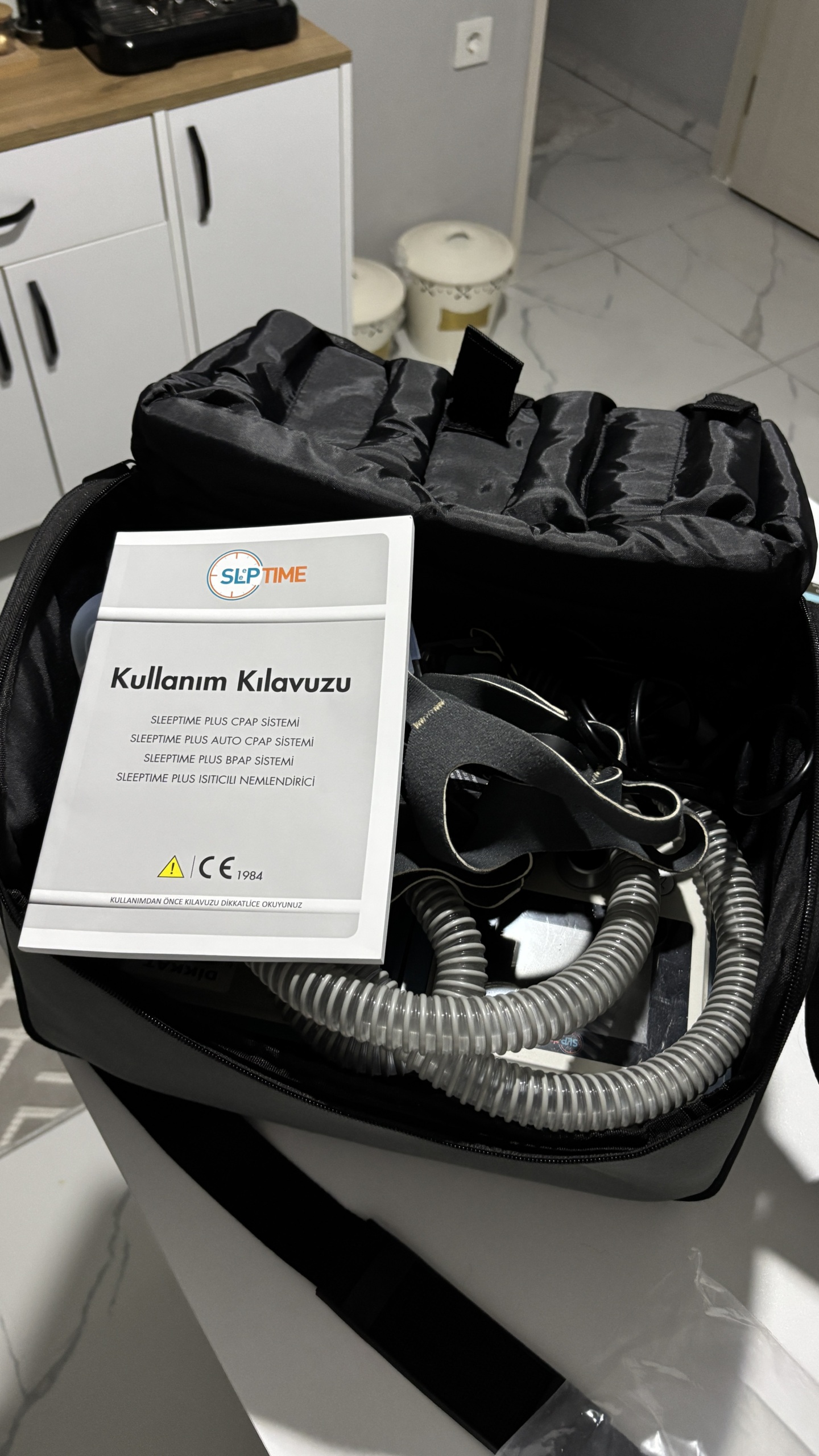
The product shown in the images is a CPAP/BPAP device used in the treatment of respiratory disorders. The set consists of the main device unit, humidifier section, breathing circuit (hose), face mask, power adapter, user manual, and carrying bag. According to the information in the user manual accompanying the product, the device belongs to the Sleptime Plus series of the Sleptime brand. The device is considered to be original.
Areas of Use
This device is used to regulate breathing and keep the airway open by providing continuous positive airway pressure during sleep, especially in conditions such as sleep apnea. It is a medical device suitable for use at home or in a clinical setting.
Quantity Information and Package Contents
According to the count made in the images, the set contents are as follows:
- 1 Sleptime Plus main device
- 1 transparent humidifier chamber attached to the device
- 1 flexible breathing hose
- 1 face mask with head strap
- 1 power adapter and electric cable
- 1 black carrying bag with shoulder strap
- 1 “User Manual” and other documents
- 1 transparent bag containing additional accessories
General Condition and State
The product has a used appearance, but its general condition is good. All main parts are present, and the device appears to have maintained its physical integrity. Slight signs of wear due to use have been observed.
Physical Deformation Examination
Upon detailed examination of the device’s physical condition, the most noticeable defect is the superficial scratches on the main unit’s screen surface. These scratches are not thought to be deep enough to affect the device’s operation. No cracks, fractures, or significant discoloration have been detected on the device casing, hose, or mask. The humidifier chamber appears transparent and clean.
Mechanical and Electronic Components
Mechanical parts such as the control knob, hose connection point, and humidifier chamber lock mechanism appear robust. Regarding the electronic components, the device is not shown operating in the images, so no comment can be made on the screen’s functionality. No visible damage is present on the power adapter and cables.
Documents and Labels
The set includes a Turkish “User Manual” containing the device’s model family and operating instructions. The “CE 1984” conformity mark is clearly legible on the manual cover. Specific technical label information such as serial number or REF code cannot be clearly distinguished on the device or other documents.
Potential Malfunction Risk Assessment
Based on the examination conducted from the images, no critical risk factor (such as rust, crushed cables, burn marks, etc.) that could lead to the device malfunctioning in the near future has been identified. The scratches on the screen are considered a cosmetic defect. The overall structure of the device and the condition of its accessories give the impression that it is in a suitable condition for regular use.