Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 19.12.2025
Respaps Respiratory Device Analysis Report
Device Identification
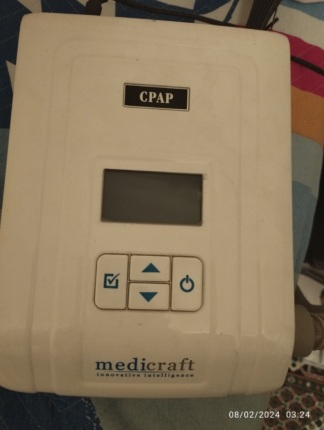
The product analyzed in the visuals is a positive airway pressure device (CPAP/BPAP derivative) used for medical purposes, typically preferred in the treatment of sleep apnea. The images show the main body of the device and the adapter unit that powers it. The product is a portable medical electronic device suitable for desktop use.
Brand and Model
On the front top panel of the device, just below the control buttons, the Respaps logo is prominently displayed. The logo is rendered with stylized lettering within a white rectangle on a black background. The overall design language of the device features the characteristic red-black color combination belonging to the Respaps brand’s product family. However, since no indication, model label, or front-face writing specifying the device’s model number (e.g., “Bipap S”, “CPAP A”, etc.) is found in the visuals, the model information could not be definitively determined.
Authenticity
Upon examining the product’s body structure, logo print quality, button layout, and screen placement, the device is evaluated to be an original Respaps production, manufactured with industrial molds and bearing brand-specific design details. It does not give the impression of a makeshift or handmade copy.
Areas of Use
This device is used to keep the airway open by providing continuous or variable positive air pressure to patients experiencing respiratory failure or cessation of breathing during sleep (sleep apnea). It is a suitable medical device for home care patients and for the pulmonology/sleep disorders specialties.
Quantity Information
A total of 2 main parts are visible in the visuals:
- 1 Red/Black Respaps Respiratory Device (Main Unit)
- 1 Power Adapter and Connection Cable
General Condition
The device is in used (second-hand) condition. There are noticeable signs of use on the product, and it is not in “new” condition. Particularly, the abrasions on the top panel indicate that the device has been actively used for a certain period and has likely been moved or frequently touched.
Physical Deformations and Cosmetic Examination
Upon detailed examination of the product’s outer casing, the following points are noticeable:
- Top Panel: The black, glossy surface of the control panel has numerous, dense hairline scratches. These scratches are clearly visible with light reflection.
- Paint and Color: There might be minor deformations or dulling due to use on the red outer frame paint in places, but no deep cracks or large breaks are observed.
- Dirt and Dust: There are accumulated dust and dirt deposits in the grooves around the control knob and along the edges of the screen. A need for cleaning is observed.
Mechanical Components
The external mechanical parts of the device appear to be in place:
- Adjustment Knob: The circular adjustment button (knob) on the top is present.
- Buttons: The two push buttons (with mute and start/navigation functions) located to the right and left of the screen appear physically intact.
- Water Chamber Cover: The black transparent cover, located on the front of the device and likely for a humidifier unit, is in a closed position.
- Air Outlet: The air outlet port on the side, used for hose connection, is open and accessible.
Electronic Components and Screen Analysis
The visuals do not provide an opportunity to test the electronic functionality of the device. The device is in an off position.
- Screen: A rectangular LCD screen is located in the center of the top panel. Since the device is not connected to power or is turned off, the screen is dark; it is not possible to ascertain from this angle whether there are pixel errors or internal screen damage. However, hairline scratches are also presumed to be present on the screen glass surface.
Accessories and Cables
Next to the device, the necessary power adapter for its operation is present:
- The adapter is black, with a round plug tip suitable for Turkey/European standards (Type C or F-like).
- The adapter cable is loosely coiled, but no broken or frayed wires are visible in its outer insulation.
- Missing Accessories: The hose (circuit) and mask, which provide air delivery to the patient, are not present in the visuals. A carrying case is not visible.
Battery Condition
It cannot be determined externally whether the device has an internal battery. However, the presence of the external power adapter next to it indicates that the device primarily operates with mains electricity. No signs of a swollen battery compartment or battery leakage were observed.
Label Information
No readable detailed technical label (Serial Number, Voltage Information, Lot Code, etc.) is found on the top surface of the device or the visible surface of the adapter. These details are typically located on the bottom of the device, and the bottom part of the device is not shown in the provided visuals. The text on the adapter is unreadable due to visual clarity.
Documents
No invoice, warranty certificate, or user manual was found in the visuals. It is unknown whether the device’s warranty is still active.
Potential Malfunction Risk
No obvious breakage or missing parts (e.g., lost buttons, compromised casing integrity) that would prevent the device from operating were observed in the visuals. However, the extensive scratches and dust accumulation on the device indicate that it may have been mishandled. Rather than a mechanical risk, the necessity for thorough hygienic cleaning and filter inspection should be considered a potential maintenance need. In second-hand medical devices, as the lifespan of the air motor cannot be determined from visuals, a “working condition” assumption should not be made based solely on external cosmetics.