Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 07.01.2026
VentMed Technology Respiratory Device Analysis Report
Device Identification
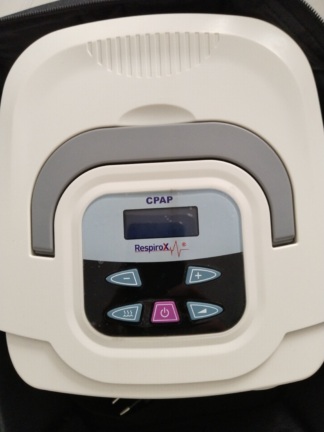
The product in the image is a medical device located inside a portable grey carrying bag. Considering the brand logo and form factor on the device, it has been determined that this product is a unit falling into the category of respiratory therapy (CPAP, BiPAP, or similar sleep apnea treatment devices). The device is positioned in the pocket of the carrying bag, with its front panel facing upwards.
Brand and Model
On the front surface of the product, there is a metallic-looking, embossed, and clearly legible logo. Based on this visual data, the brand identification is as follows:
- Brand: VENTMED TECHNOLOGY
- Model: The device’s model does not contain a clearly legible inscription due to the protective transparent packaging and the viewing angle. Therefore, a definitive determination regarding the model could not be made to avoid misleading information.
Originality Assessment
The craftsmanship of the “VENTMED TECHNOLOGY” logo on the device, the material quality, and the device’s overall casing appear to be in line with industrial standards. The details in the image give the impression that the product is an original production belonging to the brand.
Areas of Use
The VentMed brand typically manufactures devices in the field of respiratory and sleep disorder treatment. Given the structure of the device in the image, its use is probable in the following areas:
- Treatment of obstructive sleep apnea.
- Supportive treatment in cases of respiratory failure.
- Alleviation of snoring and sleep-related breathing disorders.
Quantity Information
In the image, a single (1) main device and 1 cable passing over the device are visible inside the carrying bag. No other spare parts or accessories are clearly discernible.
General Condition and Cosmetic Examination
The general condition of the device has been analyzed as follows in light of the visual data:
- Protective Packaging: The entire device is wrapped in a transparent, wrinkled protective plastic film (gelatin). This indicates that the device is either new or has been carefully packaged to prevent contamination after transport/service.
- Casing Condition: The body under the protective plastic appears clean. No visible cracks, fractures, or discoloration have been detected.
- Screen Area: There is a glossy black area at the bottom of the device, presumed to be a screen or control panel. However, due to the plastic covering, the condition of the screen (whether it is scratched or not) cannot be clearly assessed.
Physical Deformation and Malfunction Risk
During the visual inspection, no physical deformation compromising the structural integrity (dents, fragmentation, heavy impact marks) was found on the device’s casing or visible components. Due to the protective covering, potential risks appear to be minimized. However, no comment can be made regarding the mechanical functionality of the device based on the image.
Label and Service Information
On the bottom part of the device, there is a white label, partially obscured by a black cable but partly legible. This label is not a manufacturer’s factory label but is likely a technical service or distributor label. The legible parts are as follows:
- Company Name: The phrases “MEDILIFE” and “EDİKAL” (likely MEDİLİFE MEDİKAL) are discernible.
- Service Type: The letter fragments “TEKN” and “RVIS” (likely TECHNICAL SERVICE) are visible.
This label indicates that the device may have been serviced, offered for sale, or provided with service by a medical company in Turkey.
Accessories and Cables
In the image, a thin, black electrical or data cable is seen passing over the device. No apparent crushing, breakage, or deformation is visible on the cable. The grey carrying bag in which the device is located, with its zippered structure and interior texture, gives the impression of being an original accessory designed to protect the device.
Year of Manufacture and Technical Details
The device’s model label or serial number label indicating the manufacturing date is not visible from this angle. Therefore, information regarding the year of manufacture or specific technical power values (voltage, pressure range, etc.) cannot be obtained from the image.