Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 19.02.2026
MUKA Brand Electronic Intensive Care and Patient Bed Analysis Report
Device Identification and Product Description
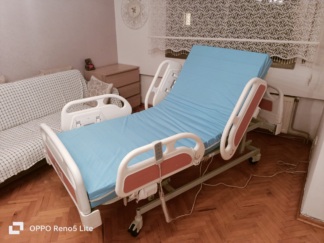
The examined visuals belong to a multi-functional, electric motor-driven patient bed. The device is designed for use in hospital environments (especially intensive care units) or for professional home patient care. The product as a whole consists of; a motorized chassis, ABS plastic sleeping platforms, four-piece independent split side rails, head/foot panels, and electronic control units.
Brand and Model Information
All branding areas and labels on the visuals have been thoroughly examined:
- Patient Bed Brand-Model: The MUKA logo is clearly legible on the product’s chassis, nurse control panel, hand remote, and integrated side rail control panels. The button combinations on the product’s control panels (Back, Leg, Height, and Trendelenburg movements) confirm that the device is a 4-motor, fully equipped model. Therefore, the product’s brand has been definitively identified as MUKA. Since the model name label is not clearly visible in the visuals, a speculative model name has not been provided.
- Patient Mattress Brand-Model: A patient mattress (bed) made of sponge or visco material is not present on the bed in the visuals. The visible beige surfaces are the bed’s own ABS plastic platform parts.
Authenticity Status
Upon examining the placement of logos on the device, the unique design of the control panels (MUKA-specific iconography and color coding), and the chassis structure, the product is assessed to be an original MUKA production. No findings indicating it is an aftermarket or assembled part have been encountered.
Control Units and Electronic Components
The product’s electronic equipment and control systems are visible in detail in the visuals. The device can be controlled from three different points:
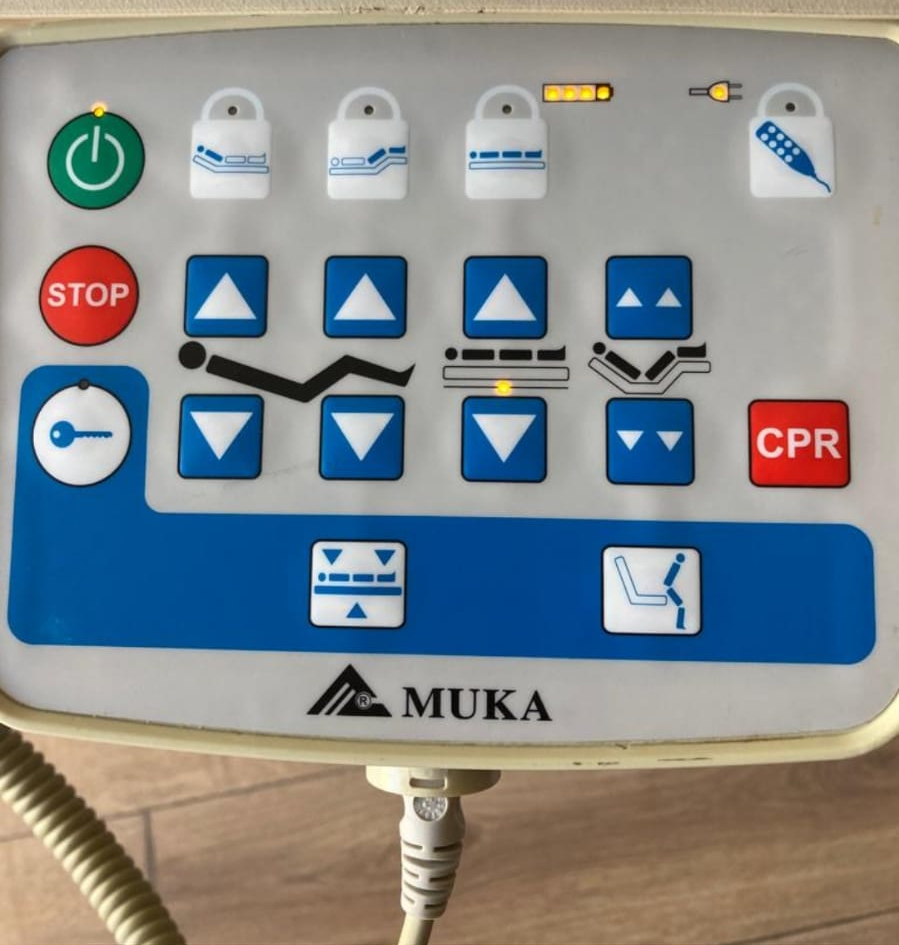
- Nurse Control Panel (Visual 3): This is the main control unit located at the foot end of the bed. This panel features “STOP” (emergency stop), “CPR” (one-button emergency intervention position), function lock keys (for the nurse to restrict patient use), Trendelenburg and Reverse-Trendelenburg movement keys, a battery charge indicator, and power indicators. The keypad is membrane type.
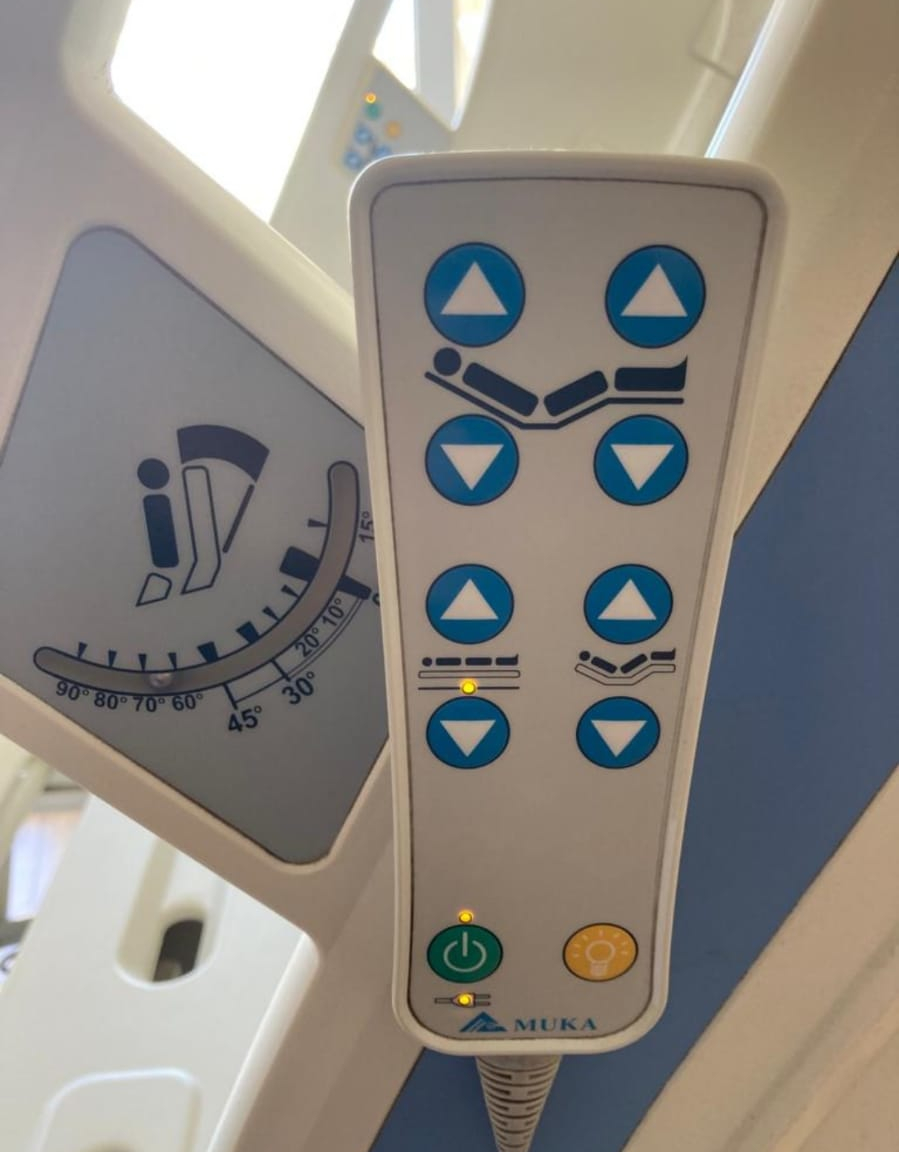
- Hand Remote (Visual 4): This is a wired unit that allows the patient to control basic functions (back elevation, leg elevation, height adjustment, and night light) from their lying position. It features the MUKA logo and function LED lights.
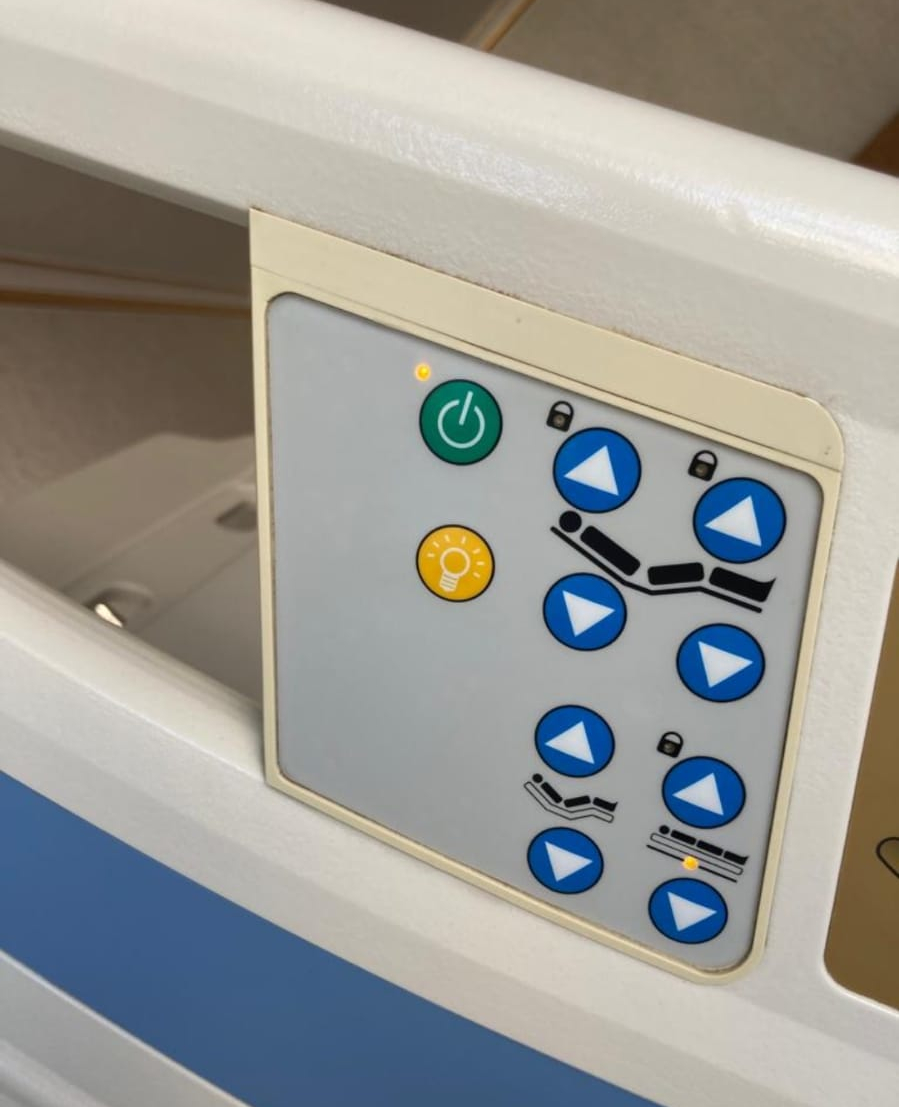
- Side Rail Control Panel (Visual 5): This is a membrane keypad integrated into the side rails, accessible by both the patient and healthcare personnel. The under-bed lighting button and movement keys are also present here.
Battery and Power Status
On the Nurse Control Panel (Visual 3), there is a battery icon accompanied by yellow LED indicators. These indicators suggest the presence of a backup battery system (accumulator) within the device to protect against power outages. However, the battery’s charge retention capacity or lifespan cannot be determined from the visual.
Mechanical Components and Physical Condition
The physical condition of the device has been analyzed under the following headings:
- Chassis and Fairing: The white metal chassis and the plastic fairings concealing the motors appear clean. No noticeable rust, deep scratches, or dents have been detected.
- Sleeping Platform: The beige ABS plastic parts forming the bed base are sturdy, the ventilation holes are open, and no broken parts are visible. Anti-slip mattress retainers are present.
- Side Rails: There are 4 independently operating shock-absorbing side rails. The angle indicators on the side rails manually show the degree of back elevation.
- Wheels: The wheel system features a stylish covered (karene) design. Based on the visuals, they appear to be in clean condition.
Deformation and Flaw Analysis
Overall, the product appears quite clean and in “good condition.” The noticeable minor details are as follows:
- On the label panels on the side rails (edges of Visuals 5 and 3), slight yellowing of the adhesive due to time or very minor lifting at the panel edges is normal, but there is no fading affecting the legibility or functionality of the buttons.
- No significant impact marks have been observed on the metal components of the device.
Areas of Use and Purpose
This product is a high-technology Intensive Care and Clinical Patient Bed. Thanks to its 4-motor structure, it can perform back, knee, height (lift), and Trendelenburg (head down/feet up) movements. It offers maximum comfort and safety (fall-preventing barriers, night light) for bedridden patients at home. Furthermore, it is preferred in hospital intensive care units due to its suitability for medical procedures (CPR, drainage positions).
Accessories and Missing Parts Check
- 1 Unit IV Pole (Stainless steel, attached to the head section).
- 1 Unit Wired Hand Remote.
- 4 Units Corner Bumper Wheels (Anti-collision).
- Missing Item: The device’s mattress (sponge bed) is not present in the visuals. The buyer will need to externally provide a mattress suitable for the bed dimensions.
Quantity Information
A total of 1 (one) unit of MUKA brand patient bed and its integrated accessories (side rails, remotes, IV pole) are visible in the visuals.
Overall Condition Assessment
The examined MUKA brand electronic patient bed appears to be in very good and well-maintained cosmetic and functional condition. Its non-yellowed plastic components and sturdy metal structure indicate that the device has either been lightly used or well preserved. The presence of critical intensive care features such as CPR, lock, and battery proves that the product is a professional medical device rather than a standard home-type bed. There is no visual evidence of mechanical failure risk (broken, detached cable, bent chassis).