Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 30.09.2025
Edan M3A Vital Signs Monitor Analysis Report
Overview and Product Description
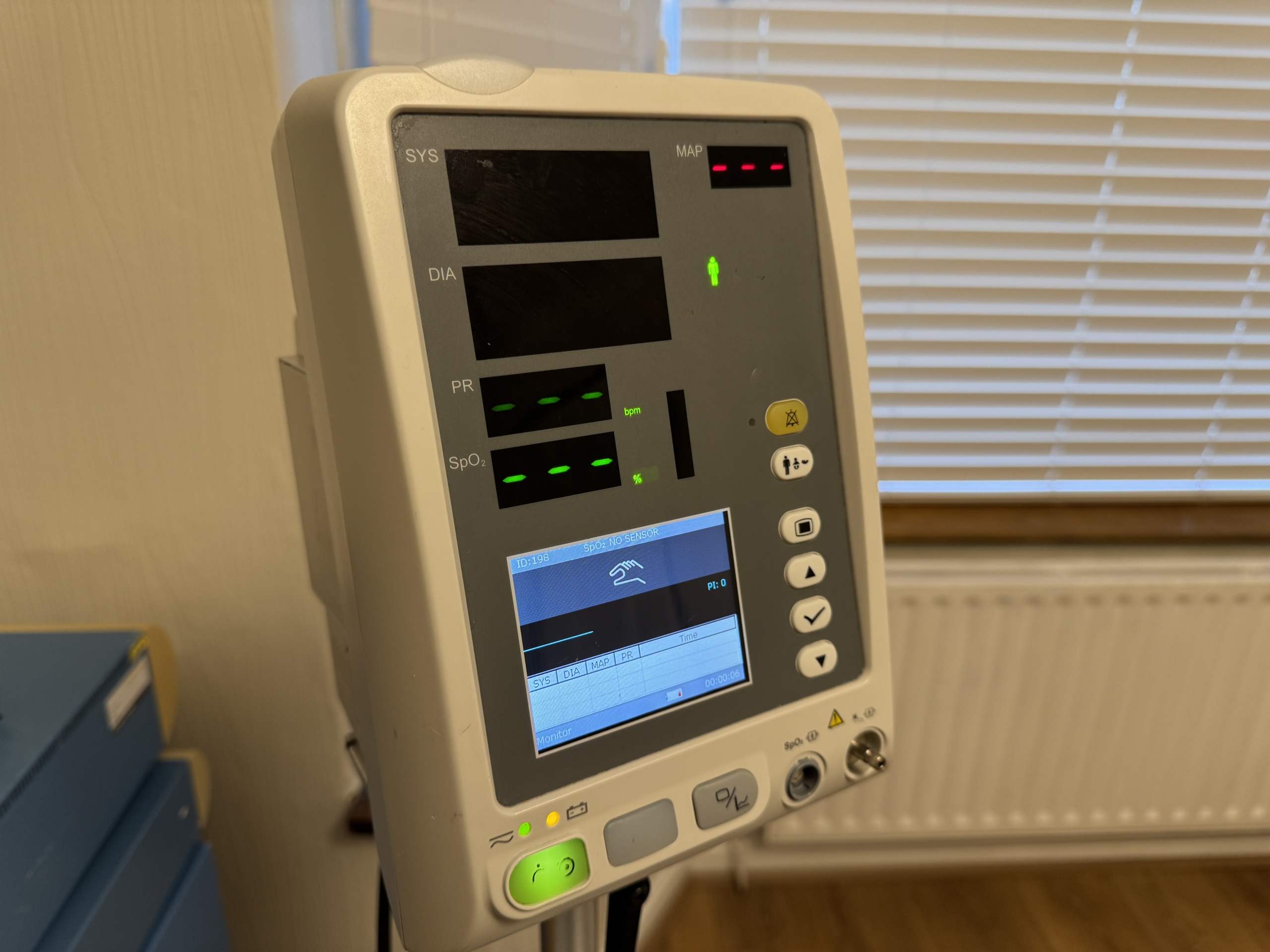
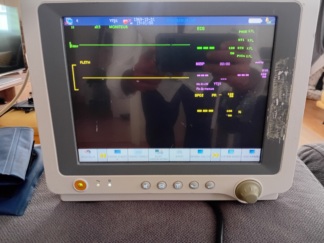
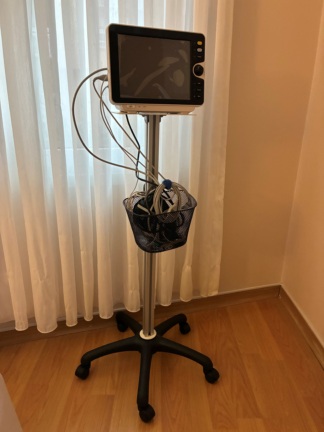
The images show a medical device mounted on a wheeled stand. According to the information on the device’s rear label, the product is an Edan M3A model vital signs monitor. The logos on the device, label quality, and overall craftsmanship suggest that the product is original. These types of monitors are used in healthcare facilities or home care settings to measure patients’ basic vital signs such as blood pressure, pulse (heart rate), and oxygen saturation (SpO2).
Quantity Information and Accessories
The products and accessories shown in the images are listed below:
- 1 unit Edan M3A Vital Signs Monitor main unit
- 1 mobile stand with basket and five wheels
- 1 blood pressure cuff and connection hose
- 1 power cable (visible inside the stand’s basket)
- 1 user manual and related documents
No missing parts were observed in the images. The device has a power input and a network (Ethernet) socket at the back.
Condition Assessment
The device is generally in good condition and is a used product. The images show the device is operational, with the screen and status lights active. Upon physical inspection, no cracks, deep scratches, or significant deformation were detected on the device’s casing or screen. There might be minor signs of use on the surfaces, but its overall cleanliness and condition are good. The mechanical components, including the wheeled stand, basket, and monitor’s mounting points, appear sturdy and functional. The control buttons on the monitor are not worn and are in place. The ventilation grille on the device’s rear panel is clean.
Technical Details and Label Information
The information on the label located at the back of the device has been examined in detail. This information confirms the device’s technical specifications and identity.
- Brand: EDAN
- Model: M3A
- Serial Number: M3112546017003090003 -01
- Production Date: 2017-05-11
The device has an internal battery. The green illumination of the battery status light on the front panel indicates that the battery is charging or fully charged. The capacity or lifespan of the battery cannot be commented on from the images, but there are no signs of physical swelling or leakage. The device’s screen is active and displays an interface showing measurement parameters (SYS, DIA, PR, SpO2). No error messages or pixel defects have been detected. The blood pressure cuff features symbols indicating its suitability for adult use.
Documents and Usage History
The “Operator’s Manual” for the product is available in the images. However, official documents such as an invoice or warranty certificate are not visible. Information regarding the device’s total usage time or its last calibration could not be obtained from the screen or labels.
Potential Failure Risk Assessment
Based on visual inspections, the device’s current condition is good. No distinct conditions posing a potential failure risk, such as crushing, fraying in the power cable, or severe damage to the casing, have been observed. The fact that the device powers on and its screen operates indicates that its basic electronic components are functional. Overall, it gives the impression of a well-maintained device.