Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 12.12.2025
Mistral-Air Patient Warming Device Analysis Report
Device Identification and Description
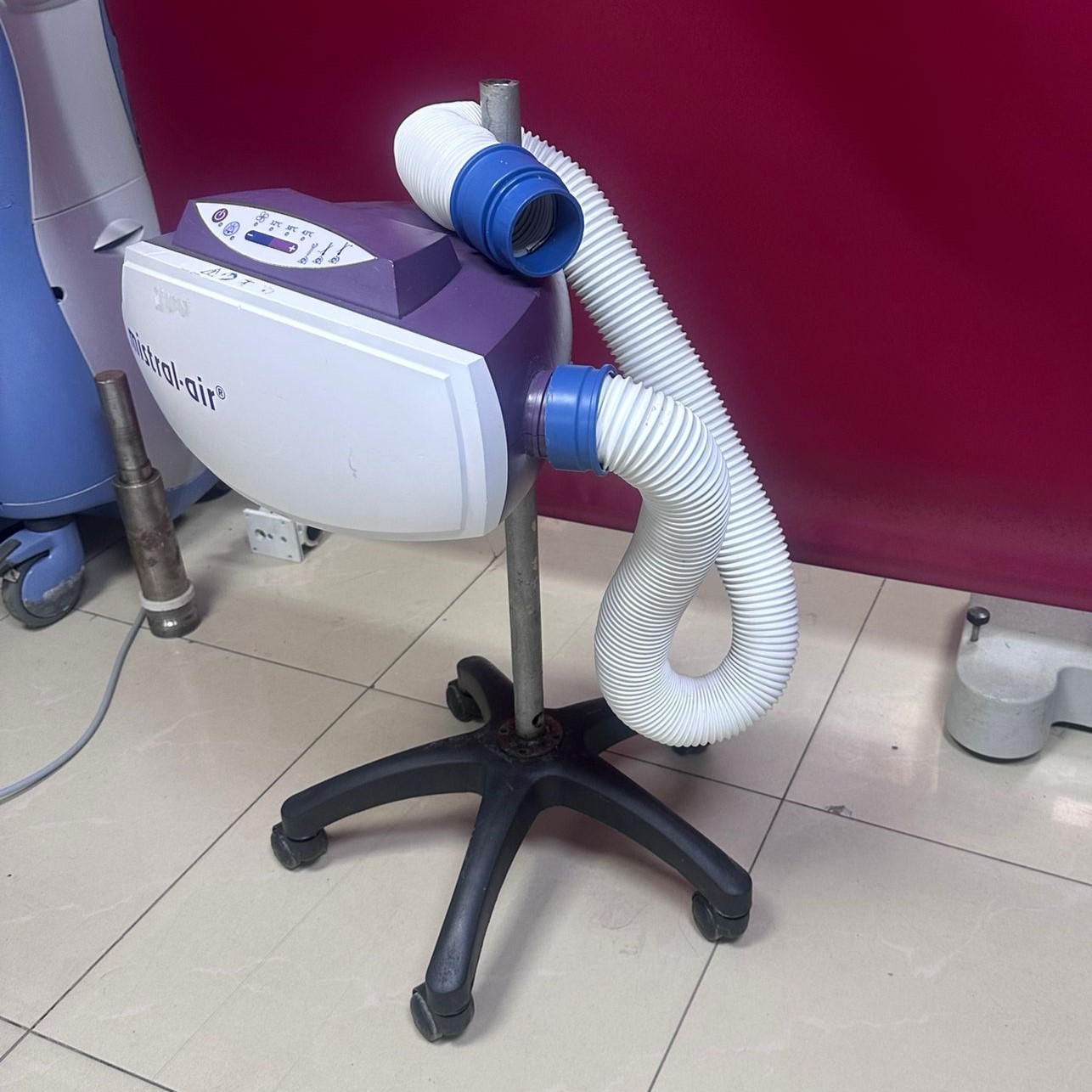
The device examined in the visuals is a professional “forced air warming system” (Forced Air Warming Unit) used in medical environments. This device is designed to maintain patients’ body temperature stably to prevent or treat hypothermia. The device consists of a main unit, an air blowing hose connected to this unit, and a mobile stand with wheels.
Brand and Model Information
As a result of detailed examination of the product’s front face and side profile, the brand information has been clearly identified.
- Brand: Mistral-Air
- Model: The general design language and panel structure of the device are consistent with standard models of the Mistral-Air series (manufactured by Surgical Company/37Company). However, since a specific numerical model code (e.g., MA1100) label on the front was not directly legible, the model name has been recorded as the general series name, “Mistral-Air”.
Areas of Use
This device is used in operating rooms, intensive care units, and post-anesthesia care units (recovery). The device provides controlled warm air through the hose, whose end is visible in the images, to special blankets (not present in the image), thereby ensuring the patient’s body temperature is maintained or increased.
Originality Status
The logo print, font type, characteristic purple-white color combination, and control panel icons on the device indicate that it is an original Mistral-Air medical device.
General Condition and Physical State
The product is second-hand and used. Although the device’s integrity is preserved, there are visible cosmetic signs of use. It is not in “zero” or “new” condition.
Physical Deformation and Wear Analysis
- Casing Surface: There is a visible label removal mark or adhesive residue on the upper left part of the front panel. Dirt and slight scuff marks are observed in places on the white plastic casing.
- Metal Components: There is a significant accumulation of rust or oxidation at the very bottom point where the metal vertical pole supporting the device joins the wheeled plastic base. The color of the metal flange part has changed and appears worn.
Control Panel and Electronic Interface
Upon examining the purple control panel on the upper part of the device, the following features were identified:
- The panel has 4 temperature setting indicators: Ambient Temperature (fan icon), 32°C, 38°C, and 43°C.
- Temperature levels are controlled with “+” and “-” buttons (Visual print).
- Additionally, a wrench and filter icons indicating a filter change warning or service requirement are present on the panel. The texts and icons on the panel are legible; there are no erasures.
Mechanical Components and Mobility
The device is mounted on a 5-arm (star base) black hard plastic or metal-blend base. Each arm has a wheel at its end. The wheels are in place and appear intact, but there is dust and dirt on the wheel covers and base parts due to use.
Accessories and Equipment Status
- Hose: A white, spiral, flexible air hose is integrated into the right side of the device.
- Hose End (Nozzle): At the end of the hose, there is a blue, hard plastic connection adapter (cuff). This part appears sturdy.
- Stand: The device is seen with its specially designed mobile stand (pole).
Quantity Information
The visuals show 1 (one) Mistral-Air warming device, along with its integrated hose and transport stand, as a complete set.
Battery Status
This type of device typically operates on mains electricity (AC 220-240V). No internal battery indicator or a battery compartment suggesting portability was observed in the visuals.
Potential Failure Risk and Assessment
As a result of the visual inspection, the rust (corrosion) at the device’s stand connection point suggests that the device may have previously been in a humid environment or exposed to liquid contact. This situation may pose a long-term risk for the structural life of the metal, but it does not appear to be a mechanical impediment for immediate use. Apart from this, no breaks, cracks, or detachments of a size that would prevent the device from operating were detected on the outer casing, hose, or panel.