Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 11.09.2025
Pulse Oximeter Device Analysis Report
Overview and Product Description
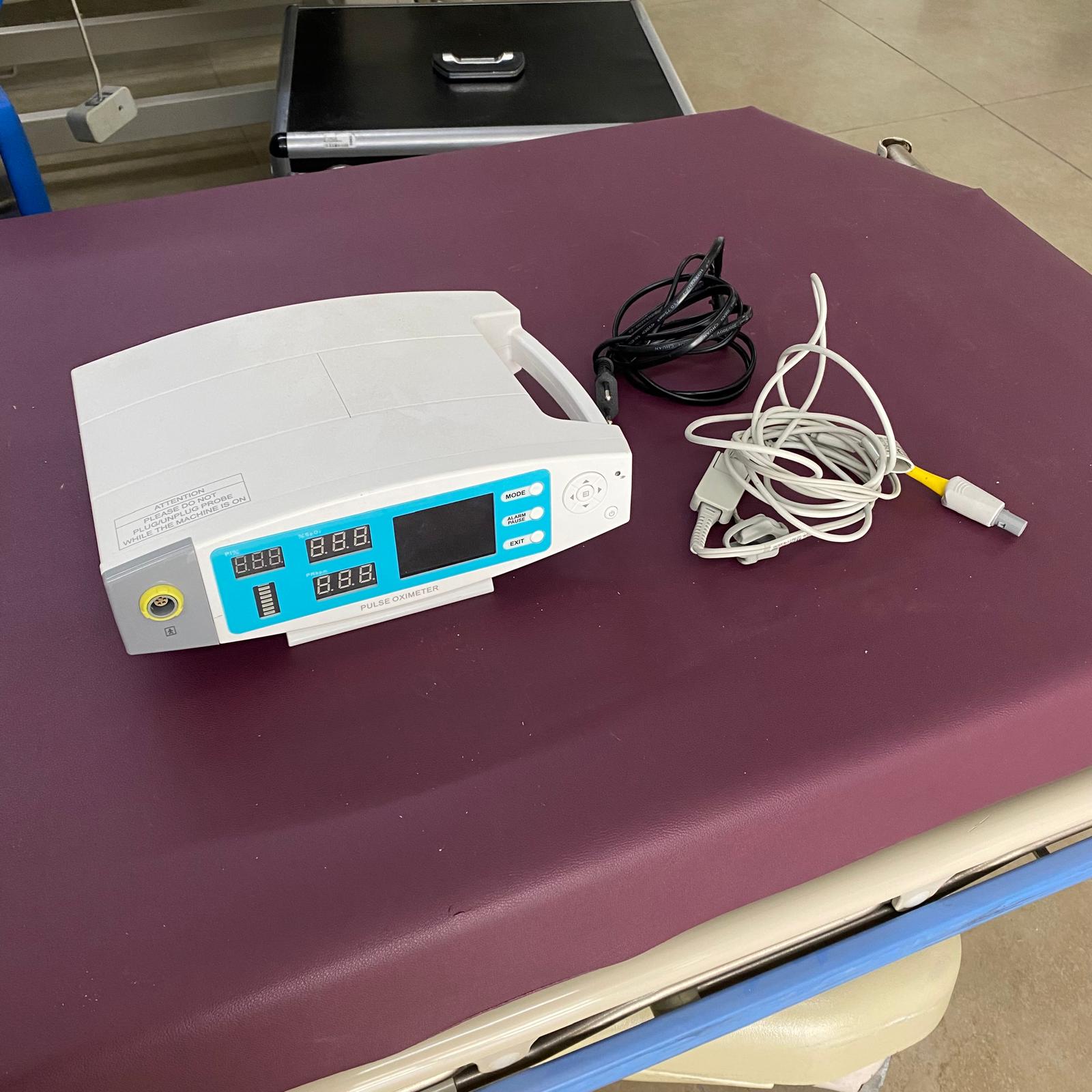
The device shown in the visuals is identified as a Pulse Oximeter. This information is clearly understood from the “PULSE OXIMETER” text on the front panel of the device. The device is used in medical settings or at home to measure a patient’s blood oxygen saturation (%SpO2) and pulse rate (PR). The general structure of the product and the sensor next to it indicate that it is a medical monitoring device.
During a detailed examination, no brand logo or model name was found on the device. Therefore, brand and model information has not been included in the report.
Quantity Information and Accessories
A total of 3 items are present in the visuals. These items are as follows:
- 1 Pulse Oximeter main unit
- 1 SpO2 sensor probe (white cable and finger clip)
- 1 power cable (black)
The device has a yellow-colored socket input for the sensor connection and an input for the power cable. It is unclear whether there are any missing accessories.
Physical Condition and Assessment
The general condition of the device is assessed as lightly used and clean. No significant scratches, cracks, dents, or discoloration were observed on its white plastic casing. The buttons, screen surface, and connection sockets on the device are in good physical condition. No signs of wear, breakage, or crushing were found on the accessory cables and sensor either.
Technical Details and Observations
Based on the examinations in the visuals, the following technical details have been identified:
- Screen and Indicators: The front panel of the device features separate digital LED indicators for %SpO2, PR (Pulse Rate), and PI% (Perfusion Index) values. Additionally, an LCD screen capable of displaying graphical data is present; however, this screen is blank as the device is turned off.
- Control Buttons: The panel includes function buttons such as “MODE”, “ALARM PAUSE”, “EXIT”, and navigation buttons. These buttons are used to manage the device’s settings and alarms.
- Warning Label: An English warning label is located on the front surface of the device. The label reads, “ATTENTION: PLEASE DO NOT PLUG/UNPLUG PROBE WHILE THE MACHINE IS ON”. No other labels containing information such as serial number, REF code, or lot number were observed.
Potential Risk Assessment
During the examination based on the visuals, no evidence of the device being faulty (e.g., broken parts, error codes on the screen) was found. The device’s casing, cables, and connectors are in very good physical condition. No serious wear, rust, or crushed cables that could pose a potential fault risk have been observed. The current condition of the device indicates a potential for problem-free operation.