Ready For Sale
Secondhand Elekta Linac Linear Accelerator
Price: USD$ 9.500,00 Approx: 427.500,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 20.03.2026
Report code: 1774002684
Elekta Precise Radiotherapy Device and Components Analysis Report
Device Identification and Areas of Use
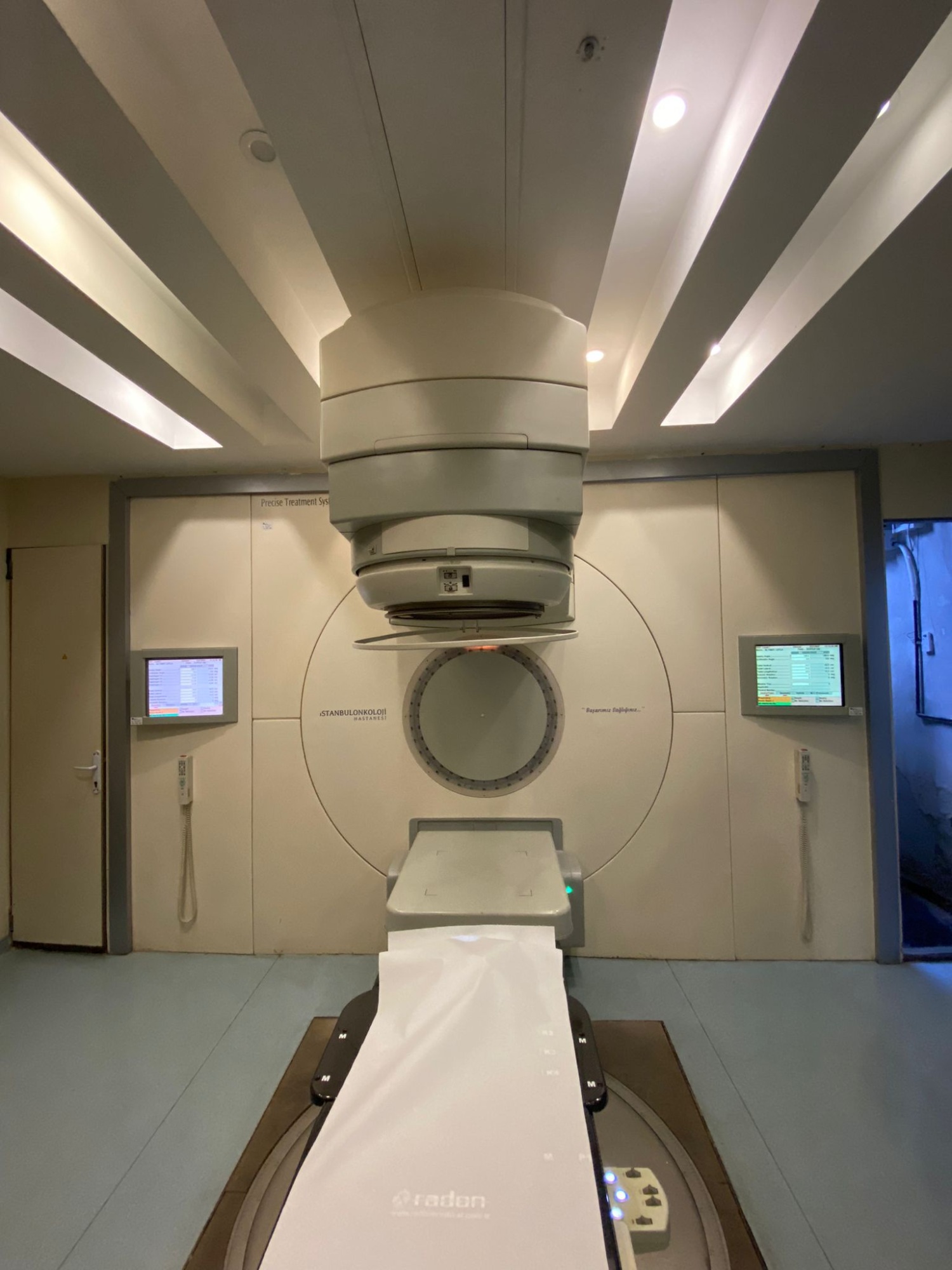
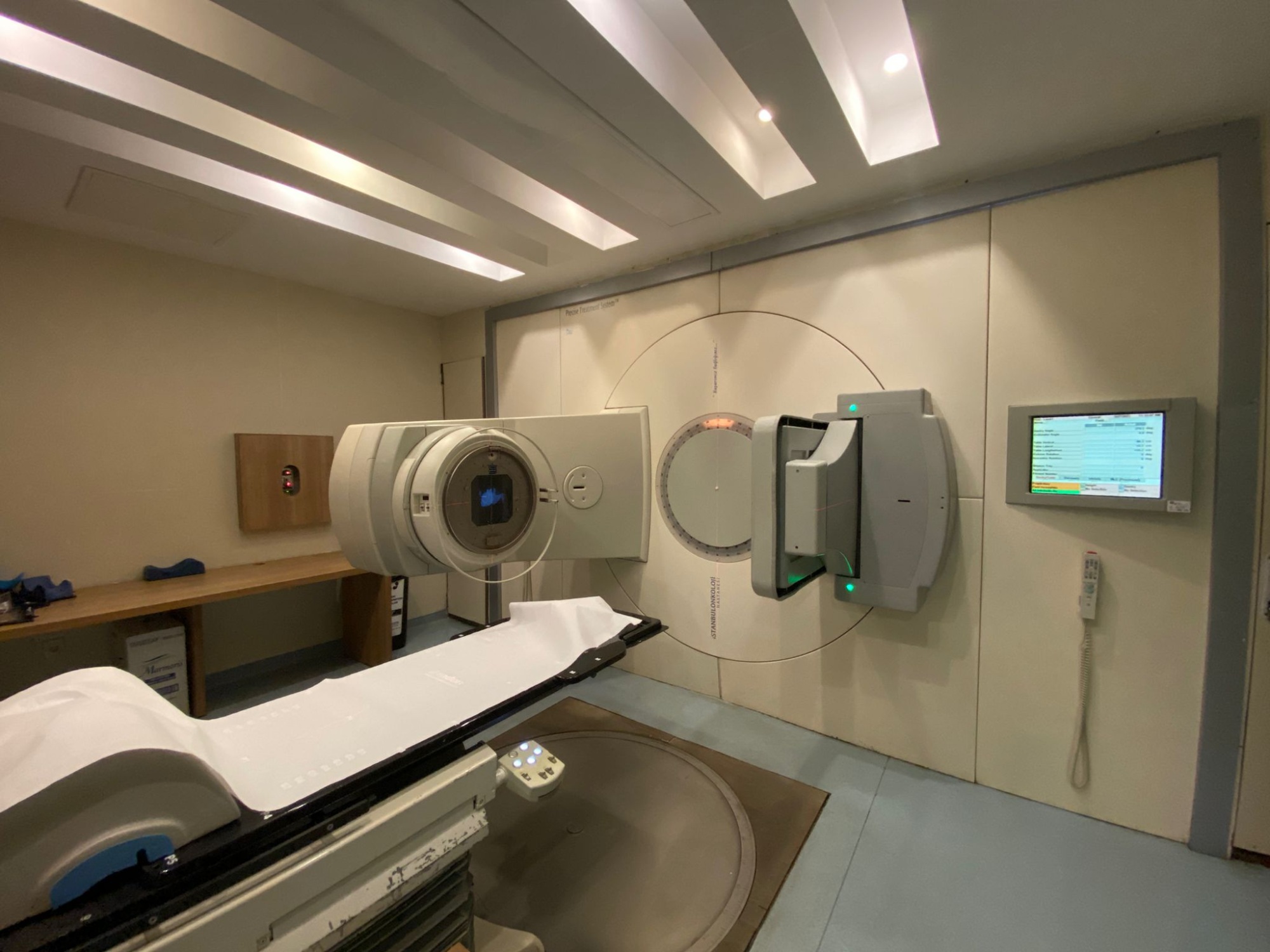
The products in the images are a linear accelerator (LINAC) radiotherapy system used for cancer treatment in the field of radiation oncology, and various disassembled electronic, mechanical, and power components belonging to this system. The system is designed to administer high-energy X-ray or electron therapy to patients. The disassembled parts can be considered as spare parts or maintenance modules for compatible devices.
Brand and Model Information
Based on the labels in the images and the inscriptions on the device, the brand of the main system and its components has been identified as Elekta, and the model as Precise. The brand Radon is visible on the underside of the patient couch.
Originality Assessment
Upon examining the general structure of the device, the heavy industrial-grade metal alloys used, custom-made RF (radio frequency) waveguides, high-voltage components, and the manufacturer labels on them, the products are observed to be original medical equipment.
Quantity and Content Information
The main units and parts identified in the images are listed below
- 1 main radiotherapy room system including installed gantry, collimator, and patient couch
- 1 disassembled Motor Drive PSU (Power Supply Unit)
- 1 high-voltage resistor and tube module
- 1 reflector module fixed and stretch-wrapped onto a wooden pallet
- 1 mechanical module containing an RF waveguide and copper cooling components
- 1 white cabinet, HP Z800 workstation, and two pulse transformers carried on a wooden pallet
General Condition and State
The installed main system is in used condition, and its general state maintains its integrity. The other disassembled components have likely undergone a dismantling process and are in the storage or transfer phase. Some of the disassembled parts are wrapped with bubble wrap and stretch film, and secured to pallets with transport straps.
Physical and Mechanical Component Analysis
The outer plastic and metal casing of the main device shows superficial traces due to use. No broken or missing outer cover has been observed. Natural oxidation (discoloration) is visible on the copper RF components among the disassembled parts. There are no dents or bends in the waveguide tubes. The outer surface of the white cabinet on the pallet has scratches and dirt caused by friction.
Electronic Component Status
The large capacitors, heatsinks, and terminal connections on the motor drive power supply (PSU) appear physically intact. The ceramic/glass tube and large green resistors in the high-voltage module are unbroken. No signs of melting or burning have been detected in the cable insulation. No batteries or battery cells are present in the images.
Screens and Accessories
There are two LCD information screens on the walls of the treatment room. The software interface showing treatment parameters is open on the screens, but the texts are unreadable due to the distant shot. Two wired hand pendants are hanging on the walls. An HP Z800 model case, which is the system’s control computer, is located on the pallet.
Label Information
The inscriptions and label information identified on the device and parts are as follows
- Wall Inscription Precise Treatment System
- Manufacturer Label ELEKTA LIMITED
- Part Description MOTOR DRIVE PSU
- Type Number TYPE No. 4513 330 7705
- Place of Manufacture MADE IN ENGLAND
- Reflector Label REFLECTORS – 1007292
- Transformer Label PULSE TRANSFORMER
- Handwritten Labels GUN RESISTOR, R4
- Cabinet Label 151532
- Connection Labels D1 – VE, D1 + VE
Potential Failure Risk
No clear breakage or burn that would prevent the device from operating has been detected in the images. However, the exposed electronic boards and high-voltage components may pose risks due to dust accumulation and static electricity. While superficial oxidation on copper parts is considered normal, it may require corrosion control at connection points. Overall, the physical condition of the parts is good.