Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 03.02.2026
GE CASE Stress ECG System and eBike Ergometer Analysis Report
Device Identification
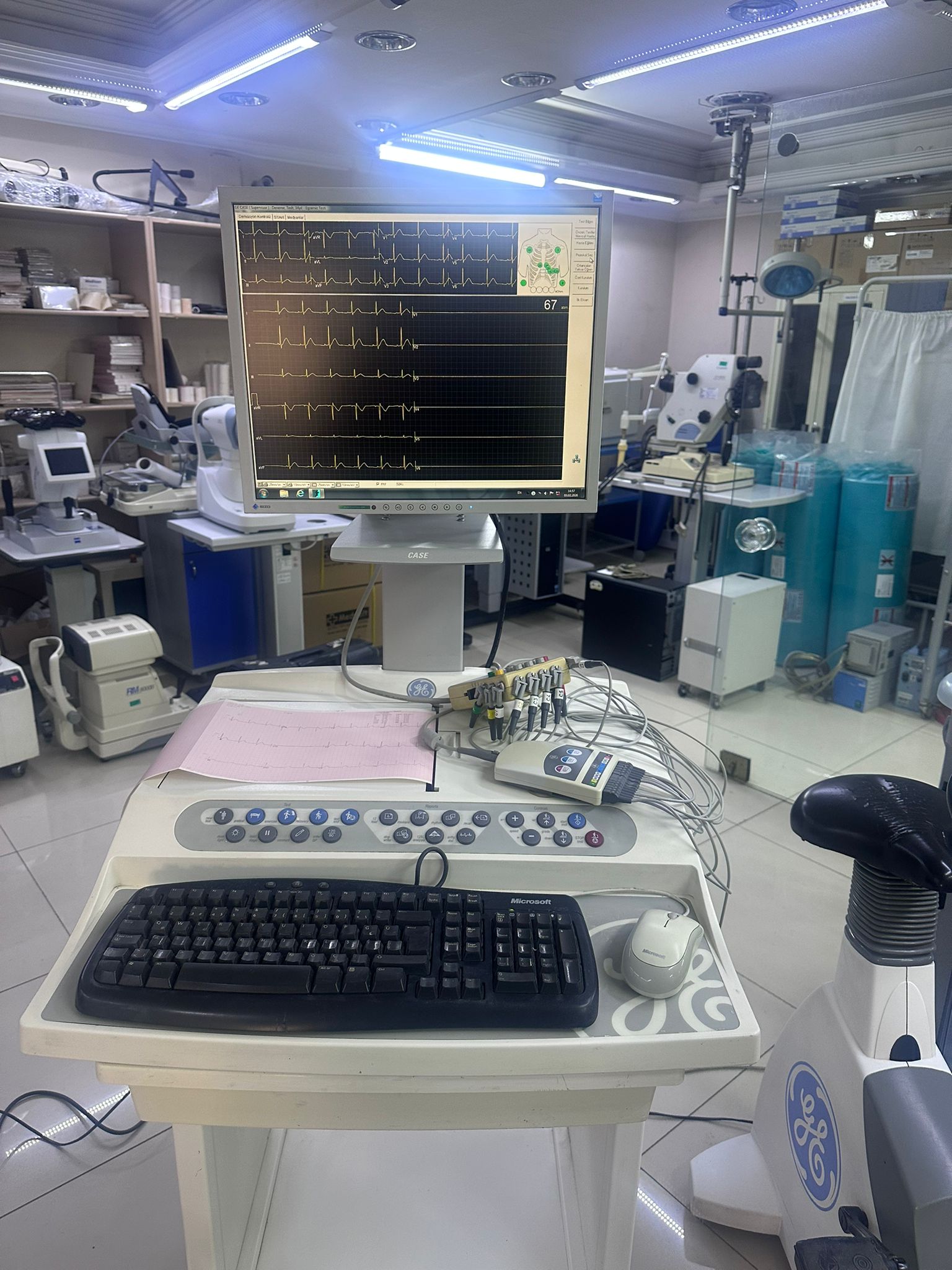
The visuals show two main medical device units working in an integrated manner. The first unit is a Stress ECG Workstation consisting of a monitor, keyboard, printer module, and a mobile stand. The second unit is a vertical type Medical Bicycle Ergometer that works in conjunction with this system and is used in patient tests.
Brand and Model
The following identifications were made by examining the logo, text, and software interface details on the visuals:
- ECG Workstation: The inscription “CASE” is clearly visible on the lower bezel of the monitor, and “GE CASE” is clearly visible in the software interface at the top left corner of the screen. This system belongs to the GE (General Electric) brand, and its model is identified as CASE.
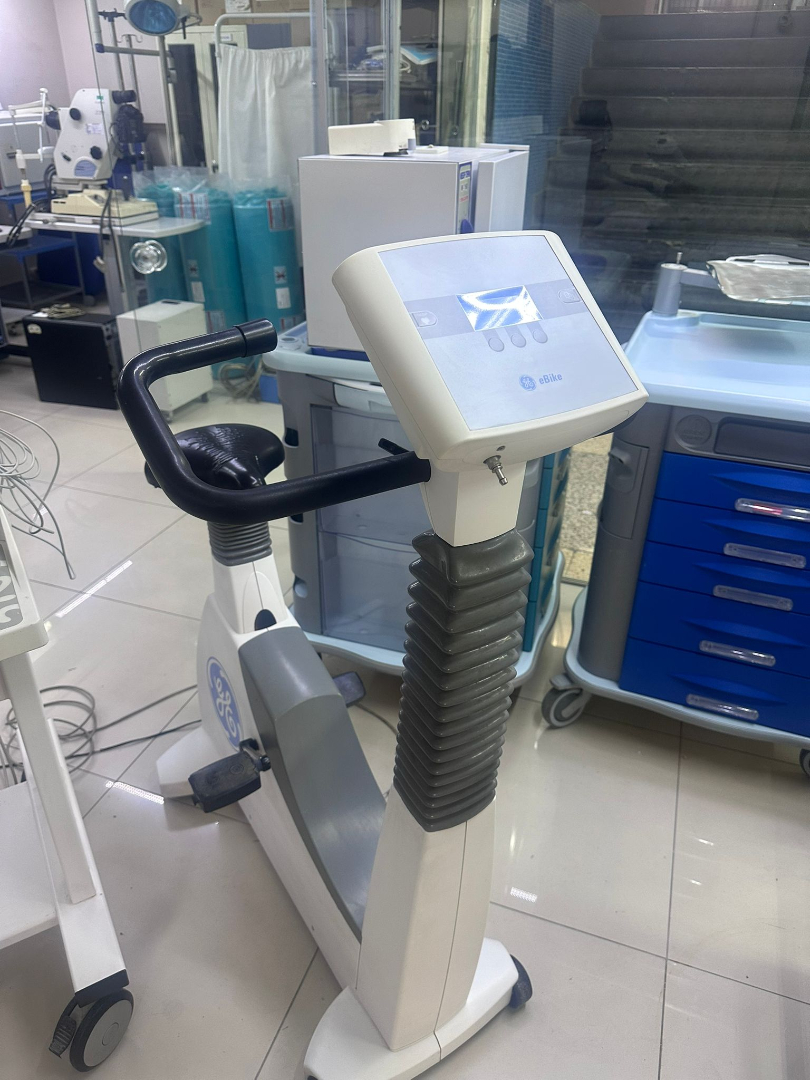
- Ergometer (Bicycle): The GE logo is present on the side body of the device and on the control panel. The model name eBike is clearly readable on the control head. The brand is GE, and the model is eBike.
- Keyboard: The Microsoft brand is visible on the keyboard connected to the workstation.
Originality
The placement of the logos on the devices, GE’s characteristic color tones (white/gray and blue details), the licensed appearance of the interface software, and the material quality indicate that the products are original GE Healthcare productions.
Areas of Use
This system is used in cardiology clinics and hospitals to perform a procedure known as “Stress Test.” The bicycle (ergometer) allows the patient to exert effort in a controlled manner, while the CASE station continuously monitors, records, and analyzes the patient’s ECG data. It is suitable for use in the diagnosis of cardiovascular diseases, analysis of arrhythmia, and monitoring of treatment.
Quantity Information
The visuals show a total of 2 main units: 1 (one) GE CASE ECG system (including trolley/stand, monitor, processor unit) and 1 (one) GE eBike ergometer bicycle. Additionally, there is one ECG cable module (patient interface), one keyboard, and one mouse.
General Condition
The devices are second-hand, used. However, their overall cosmetic condition appears clean and well-maintained. The devices are shown in working condition; there is an active signal flow on the monitor and illumination on the bicycle screen.
Physical Deformations
During the detailed visual inspection:
- Ergometer Screen: As seen in the 5th visual, there are slight bulges or superficial undulations on the screen film or coating of the eBike control unit, understood from the reflection of the screen light. However, these do not obstruct the screen display.
- Casings: No significant cracks, fractures, or yellowing were observed on the outer plastic components of either device. The white color tone has been preserved.
- Upholstery: The saddle leather of the bicycle appears intact; no tears were detected.
Mechanical Components
The corrugated gray plastic protectors (accordion-type dust covers) that provide handlebar (grip) and saddle height adjustments on the bicycle are intact and in place. The trolley wheels are complete. Bicycle pedals and foot securing straps are present.
Electronic Components
The devices were in working condition when the visuals were taken.
ECG System: The monitor is functional and displays 12-lead ECG traces instantaneously. This indicates that the processor unit and display unit are working without issues.
Bicycle Ergometer: The control panel screen is operating with blue backlighting. No pixel loss was visually detected on the LCD screen.
Accessories
The examined parts are as follows:
- ECG Module: A white-colored data collection module (Acquisition Module), where patient cables are connected, is visible on the front tray of the system. Electrode cables are attached to it.
- Thermal Printer Paper: Pink grid-patterned ECG paper is seen protruding from the internal printer located at the front of the ECG unit.
- Input Devices: A black, wired Microsoft brand Q keyboard and a standard white/gray wired mouse are connected to the system.
Battery Status
From the visuals, no technical inference can be made about the device’s internal battery status (charge retention capacity, lifespan, etc.). However, it should be assumed that the devices operate connected to mains electricity via cable.
Label Information
Labels indicating model names (eBike, CASE) and containing brand logos (GE monogram) are present on the products. However, “technical data plate” labels containing serial numbers, lot numbers, manufacturing dates, or detailed electrical values are not clearly legible from the angles in the visuals.
Screen Analysis
Main Monitor: The “GE CASE” software interface is open. Multi-channel ECG rhythm waveforms (PQRST complexes) are flowing on the screen. An electrode placement indicator (green dots) is visible on a body diagram in the upper right corner. This suggests that the device is connected to a simulator or operating in test mode.
Bicycle Screen: Parameter headings such as “Watt”, “NIBP” (Non-Invasive Blood Pressure), and “mmHg” are selected on the blue-backlit LCD screen. However, numerical values (0 or –) indicate that the test is not active.
Size and Compatibility
The ergometer is adult-sized. Since handlebar and saddle heights are adjustable, it is suitable for adult patients of various heights. The system is of a size that can be used in a standard clinical room.
Documents
No invoice, warranty certificate, calibration report, or user manual belonging to the products is present in the visuals.
Existing Faults
Based on visual evidence, no error messages (“System Error”, “Sensor Fail”, etc.) are observed on the system. The smooth flow of ECG waveforms on the screen indicates that the system’s signal processing capability is functional. No visible broken or detached parts are present.
Potential Fault Risk
As the product is second-hand, the slight deformation observed on the bicycle screen surface in the visual may pose a cosmetic issue for the keypad or screen readability over time. Although mechanical moving parts (pedal system, height adjustment) appear robust in the visual, the condition of the internal gears should be tested. Currently, no serious potential risk has been visually detected.