Ready For Sale
Secondhand F.Bosch Provac Suction Device
Price: USD$ 30,00 Approx: 1.350,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 23.02.2026
F. Bosch Medizintechnik Provac Electric Suction Device Analysis Report
Device Description and Brand Identification
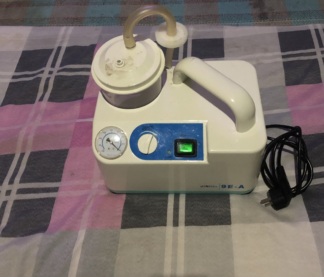
- Device Identification: The displayed device is a portable electric suction (vacuum) device used for clearing body fluids in medical procedures or home care settings.
- Brand and Model: As a result of the analysis performed on the device’s front panel and original packaging, the brand has been definitively identified as F. Bosch Medizintechnik and the model name as Provac. The phrase “Provac Elektrikli Aspiratör Cihazı” (Provac Electric Suction Device) is clearly legible on the box.
- Originality: The device’s casing craftsmanship, logo print quality, corporate fonts on the packaging, and accessory compatibility indicate that the product is an original F. Bosch production.
Areas of Use and Purpose
The product is suitable for use in ear, nose, and throat, home patient care, emergency services, and minor surgical intervention areas for clearing fluids such as mucus, phlegm, or blood from the respiratory tract. Its portable design makes it suitable for mobile healthcare services.
Quantity and Accessory Breakdown
- 1 Unit F. Bosch Provac Suction Main Unit
- 1 Unit Collection Jar (Fluid reservoir)
- 1 Unit Jar Lid (Metal/Chrome-look)
- 1 Unit Patient Connection Hose (Silicone)
- 1 Unit Device Connection Intermediate Hose
- 1 Unit Hydrophobic Filter (On the hose line)
- 1 Unit Original Product Box
Condition and Assessment
- General Condition: There is no yellowing, discoloration, or significant signs of use on the device’s outer casing. The plastic surfaces retain their shine. The product is in new or very lightly used (e.g., for demonstration purposes) condition.
- Physical Deformation: No cracks, breaks, deep scratches, or dents have been detected on the device body, control panel, or collection jar. The box lid has minor deformation and bends due to opening.
- Mechanical Components: The carrying handle is intact and integrated with the body. The pressure adjustment knob (regulator) is in place and firm. The jar lid appears to be seated correctly.
- Electronic Components: Within the scope of visual analysis, motor operating performance cannot be assessed as the device was not electrically connected. However, there is no physical damage visible on the switches and indicators from the outside.
Technical Analysis and Components
- Manometer (Pressure Gauge): A circular, analog vacuum gauge is located on the front panel. The indicator scale is for monitoring the amount of negative pressure applied. There are no cracks or fogging on its glass.
- Adjustment Knob: The chrome-detailed circular knob on the right side is used to increase or decrease the vacuum power. “+” and “-” symbols are present on the panel.
- Fluid Collection Reservoir: Made of transparent polycarbonate material. A scale line indicating the 1000 ml level is present on the reservoir. No dullness or calcification is observed in the reservoir.
- Battery Status: The phrase “Elektrikli Aspiratör” (Electric Suction Device) is on the device box. There is no indication or visual data (e.g., charge indicator) regarding an internal battery. It is assessed to operate with mains electricity.
- Technical Information on the Box:
- Medium Vacuum, Low Flow
- Oil-Free Piston Pump (Indicates a maintenance-free motor type)
- Adjustable Pressure Knob
- 2 Years Warranty (It is unknown whether the warranty period has started)
Risk and Potential Malfunction Assessment
During the visual inspection, no risk factors threatening the mechanical or structural integrity of the device were observed. There are no signs of kinking, breakage, or hardening (yellowing) in its hoses. As a potential risk, the rubber seals might need to be checked based solely on the product’s shelf life, but the current visual data indicates a clean and solid profile.