Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 02.10.2025
Laerdal LSU Aspirator Device Analysis Report
Overview and Device Description
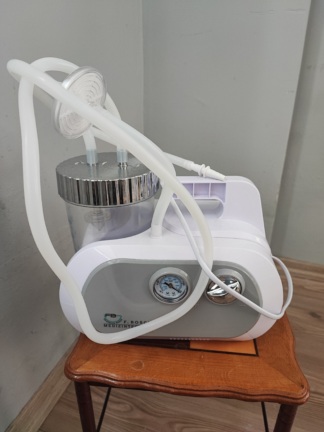
The images show a portable aspirator device intended for medical use. The device is specifically designed to clear liquids such as secretions, blood, or vomit from patients’ airways. Its compact and portable structure indicates its suitability for use in emergency situations and during patient transfers. The product features a yellow and navy blue main body, a transparent collection canister, and a control panel. The total number of units in the images consists of one main unit.
Brand and Model Information
During a detailed examination, the words Laerdal Suction Unit and LSU are clearly legible on the front of the device. Based on this information, the brand of the device has been definitively identified as Laerdal, and the model as LSU (Laerdal Suction Unit). The device’s logo and model inscriptions, combined with the overall quality of workmanship, give the impression that the product is original.
Physical Condition and Deformation Analysis
The general condition of the device is assessed as used. Superficial scratches and minor soiling, consistent with use, have been observed on the yellow and navy blue plastic sections of its casing. However, no major cracks, fractures, or dents that would affect the overall integrity of the device have been detected. No cracks or fractures are visible on the transparent collection canister either. The measurement lines (100, 700, 1000 ml) on the canister are distinct.
Mechanical and Electronic Components
Mechanically, the vacuum adjustment knob on the device’s front panel is physically in place and shows no signs of damage. The blue mechanism holding the collection canister and the metal connection ports on the canister lid also appear robust. Electronically, the device’s front panel features an LED-indicated vacuum level panel and a “TEST” button. As the device is not shown in operation in the images, no comment can be made regarding the functionality of this panel.
Accessories and Missing Parts
The images show the device’s main body and an attached 1000 ml capacity collection canister. However, essential accessories such as patient connection hoses (aspiration catheter), a charging cable, or an adapter, which are necessary for performing the aspiration procedure, are not present. It is not clearly discernible from these angles whether the device has an external power input socket, which implies that the parts required for power supply and battery charging are missing.
Labels and Technical Information
During the examination of the device, no label containing specific production information such as a serial number, REF code, or lot number was detected. However, the device’s front panel features a scale indicating vacuum power from 0 to 500+ mmHg. On its side, there is a pictorial user manual label; this label illustrates how to assemble the parts in three steps. The collection canister also has measurement lines indicating liquid volume.
Potential Risk Assessment
Although the device does not have any severe physical deformation or breakage, its performance needs to be thoroughly tested due to it being a used product. The most significant potential risk is the absence of critical accessories such as the power cable and patient hoses. Furthermore, as it is a portable unit, the current condition, lifespan, and charging capacity of its probable internal battery are unknown. It is not possible to operate and use the device without addressing these deficiencies. Nevertheless, the device’s casing and core components are visibly in good condition.