Ready For Sale
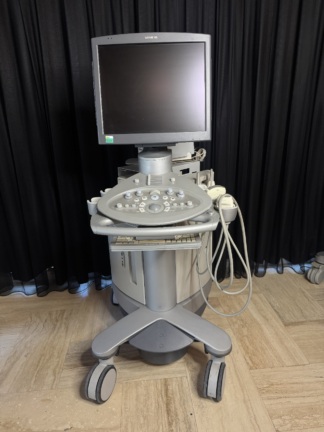
Secondhand GE Logiq E9 Ultrasound Machine
Price: USD$ 21.000,00 Approx: 945.000,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 26.11.2025
GE LOGIQ E9 Ultrasound Machine Analysis Report
Device Identification and Area of Use
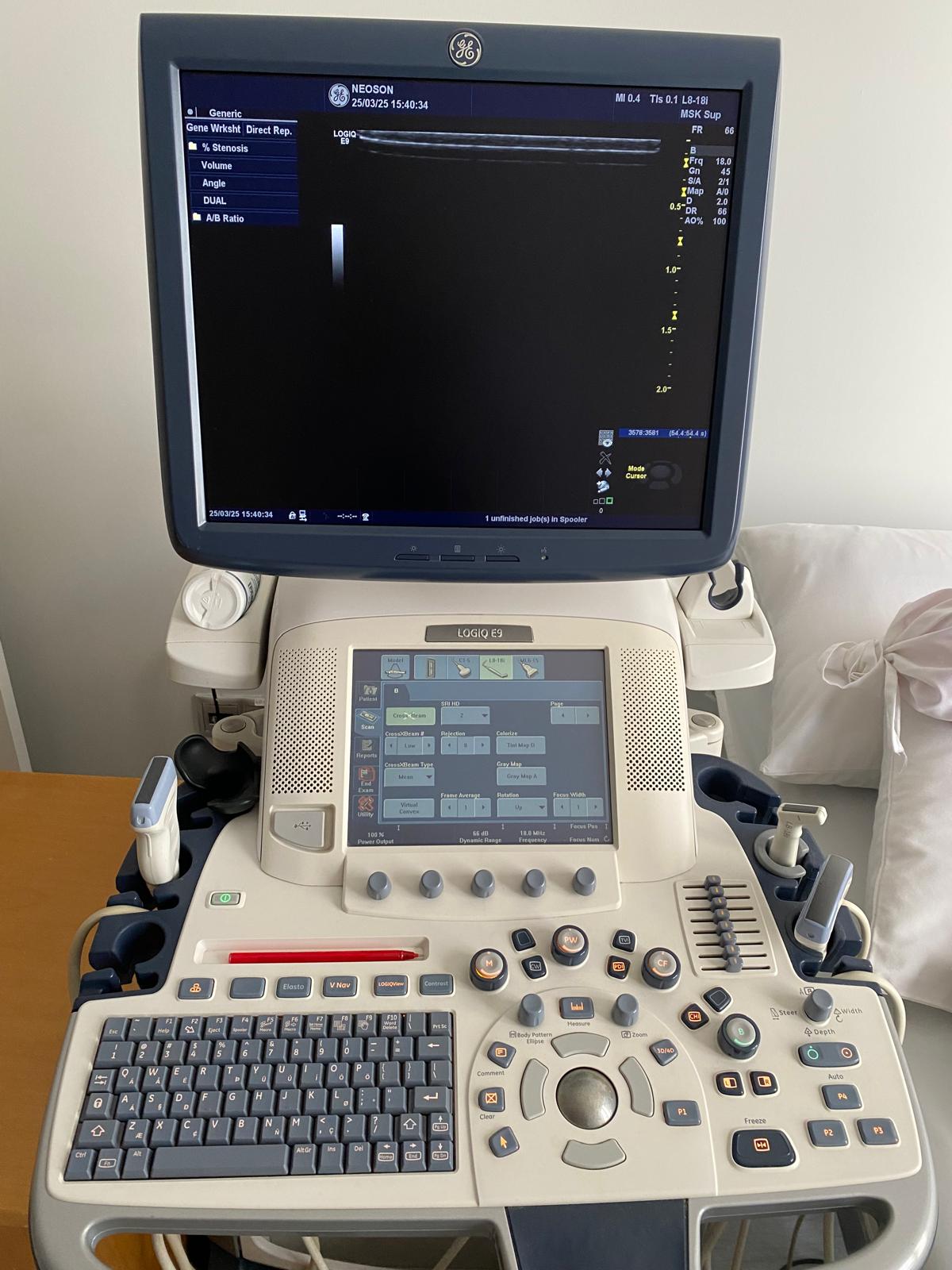
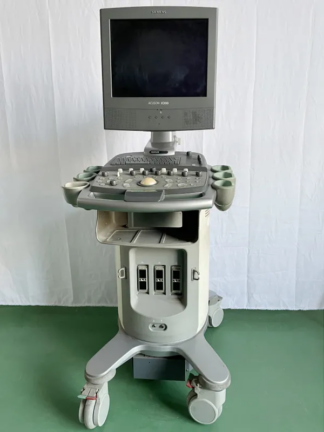
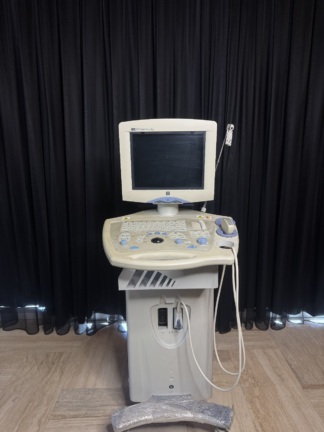
The device shown in the visuals is a high-end ultrasonography system used for medical imaging. It is understood to be used in diagnostic processes for radiology, obstetrics/gynecology (OB/GYN), vascular, cardiology, and general imaging purposes. The device is a full-featured console-type ultrasound with a wheeled stand, a large LCD monitor, a touch control panel, and an integrated keyboard.
Brand and Model Information
As a result of detailed visual analysis, the brand and model of the device are definitively as follows:
- Brand: GE (General Electric)
- Model: LOGIQ E9
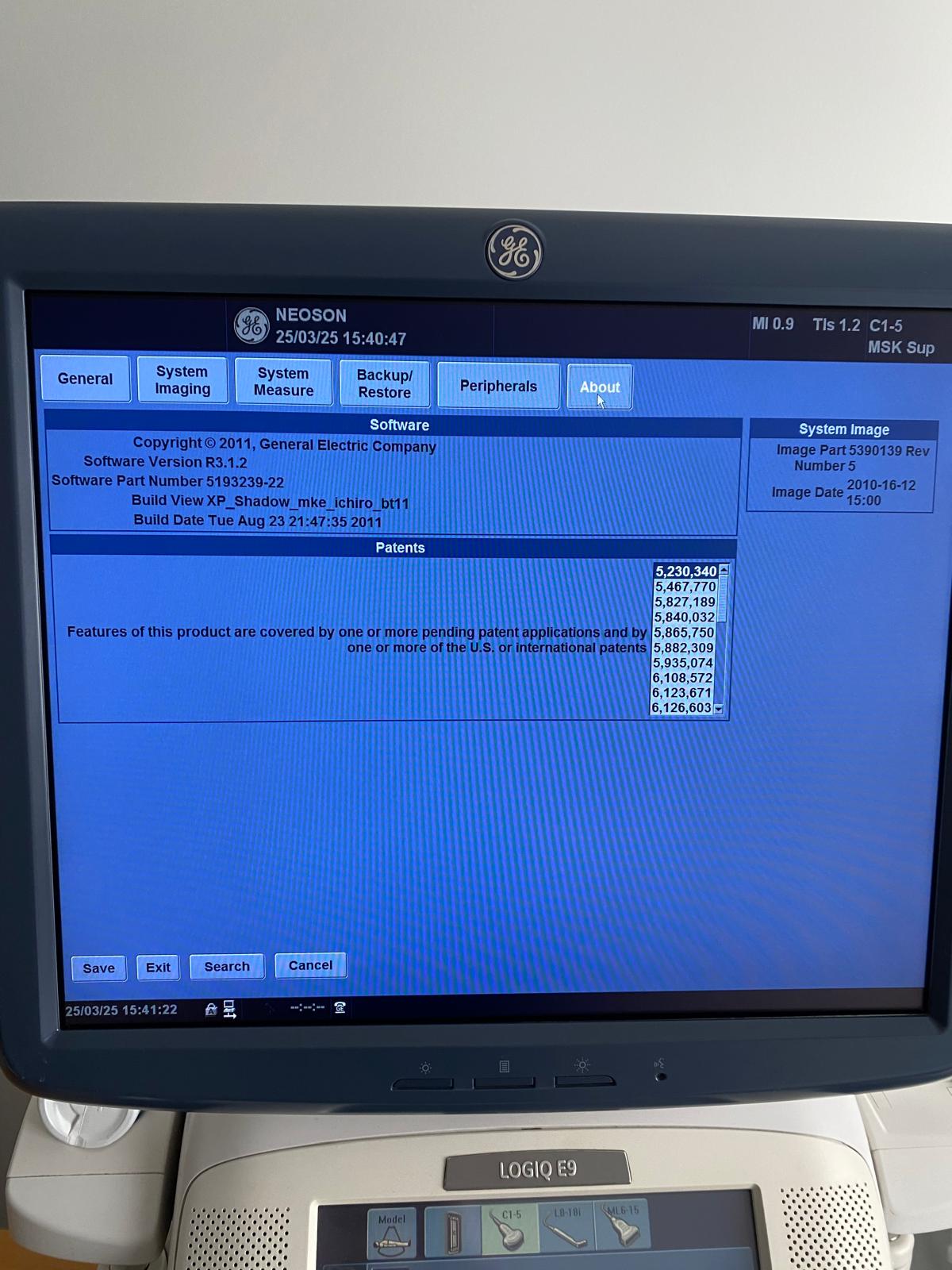
This identification was made with 100% certainty based on the “GE” logo on the upper bezel of the main screen, the “LOGIQ E9” inscription on the touch panel, “LOGIQ E9” appearing in the software titles within the screen, and the software information on the system information screen (About screen).
Hardware and Configuration Analysis (Technical Data)
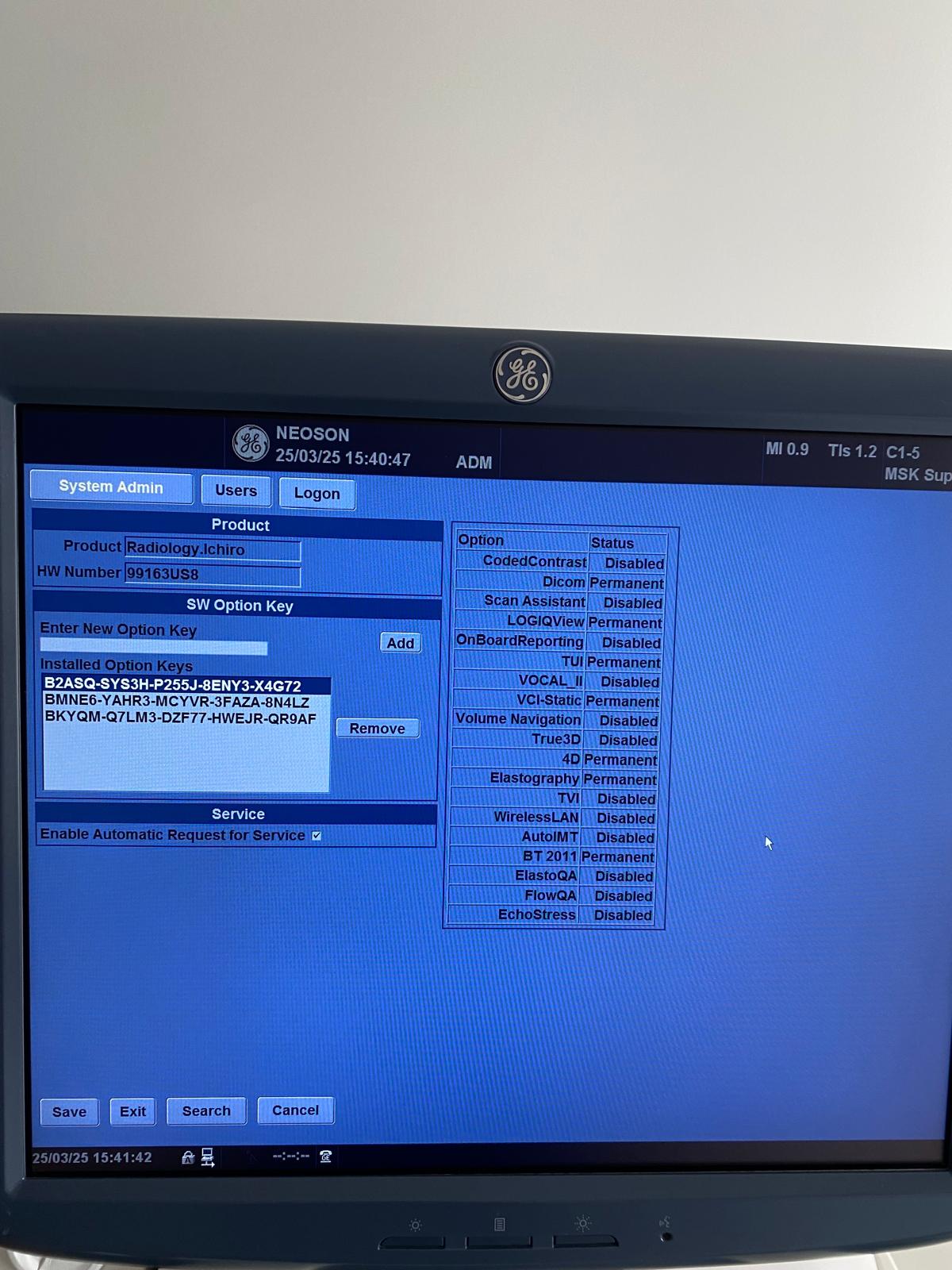
From the system information screens in the visuals, the technical history of the device and its licenses have been thoroughly analyzed. The obtained data are as follows:
- HW (Hardware) Number: 99163US8
- Software Version: R3.1.2
- Software Part Number: 5193239-22
- Build Date: 23 August 2011 (Build View: XP_Shadow_mke_ichiro_bt11)
- Copyright: 2011, General Electric Company
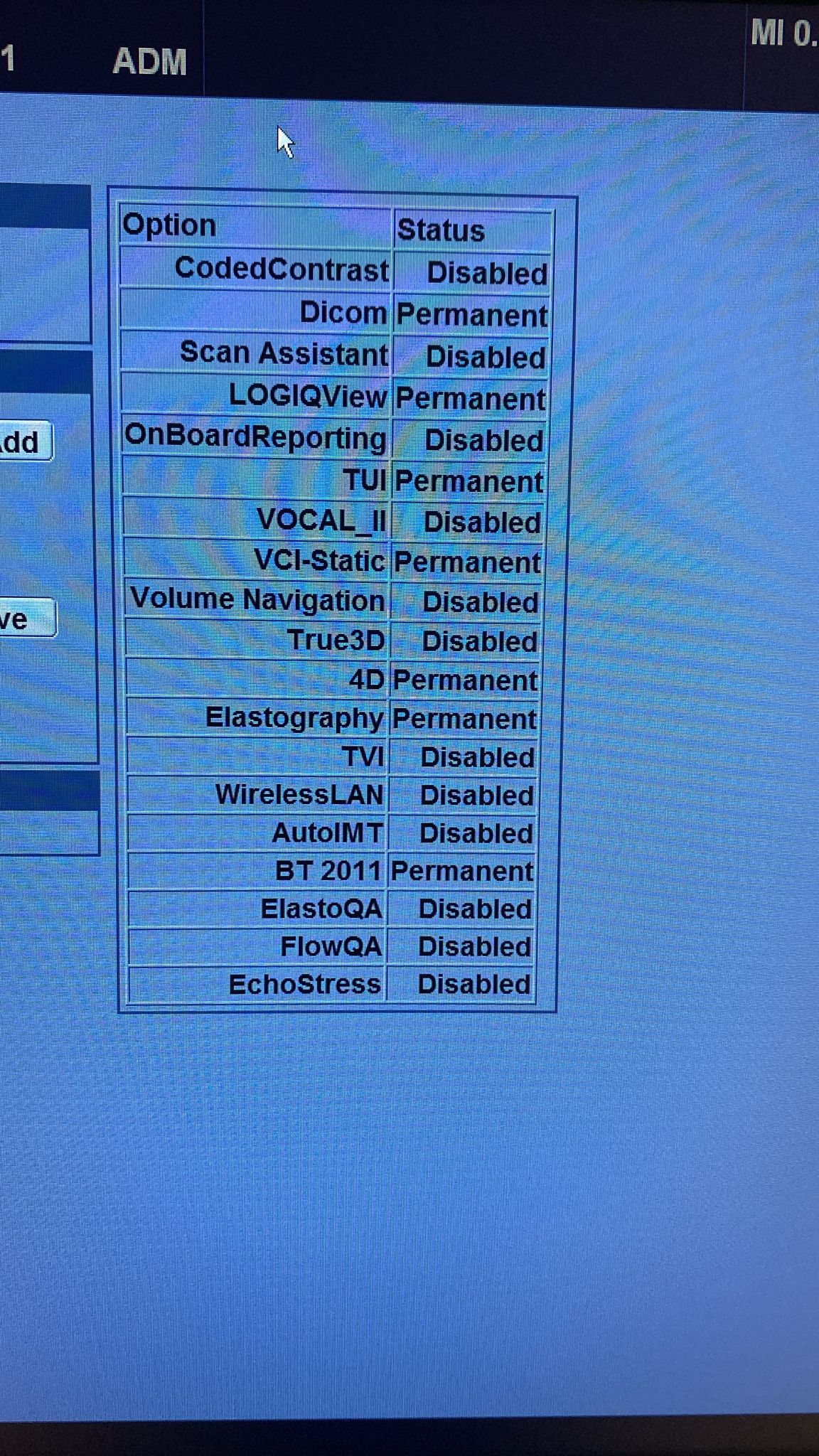
Active Options and Licenses
The following features are marked as “Permanent” in the device’s “Option” menu, and these features determine the device’s value and usage capacity:
- DICOM: Active (Image archiving and communication system compatibility).
- LOGIQView: Active (Panoramic imaging).
- TUI: Active (Tomographic Ultrasound Imaging).
- VCI-Static: Active (Volume Contrast Imaging).
- 4D: Active (Real-time 3D imaging, a critical feature particularly for obstetrics).
- Elastography: Active (Tissue stiffness measurement, important for cancer screenings).
- BT 2011: Active.
Some features such as WirelessLAN, Scan Assistant, and True3D are observed to be “Disabled”.
Accessory and Probe Analysis
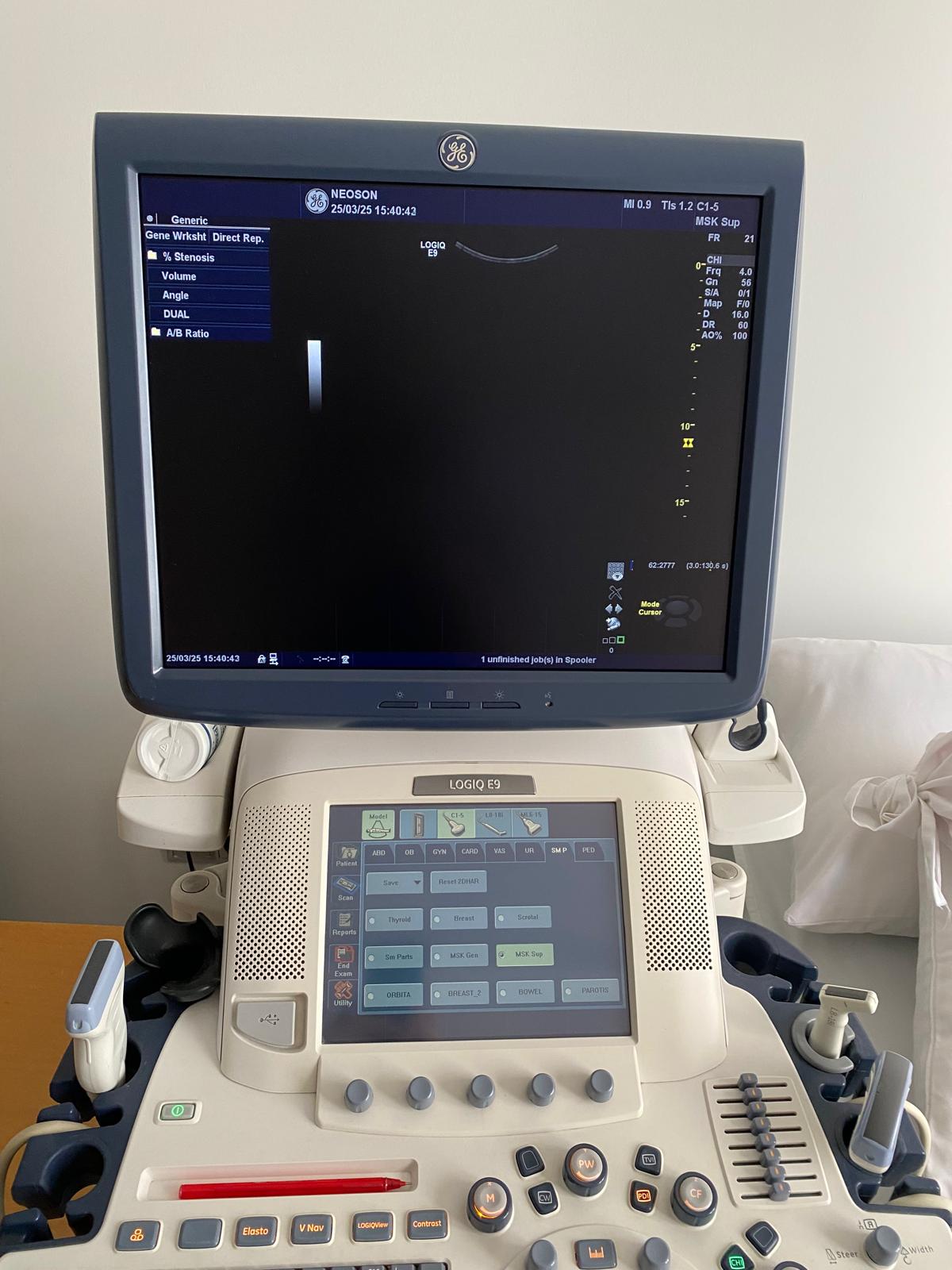
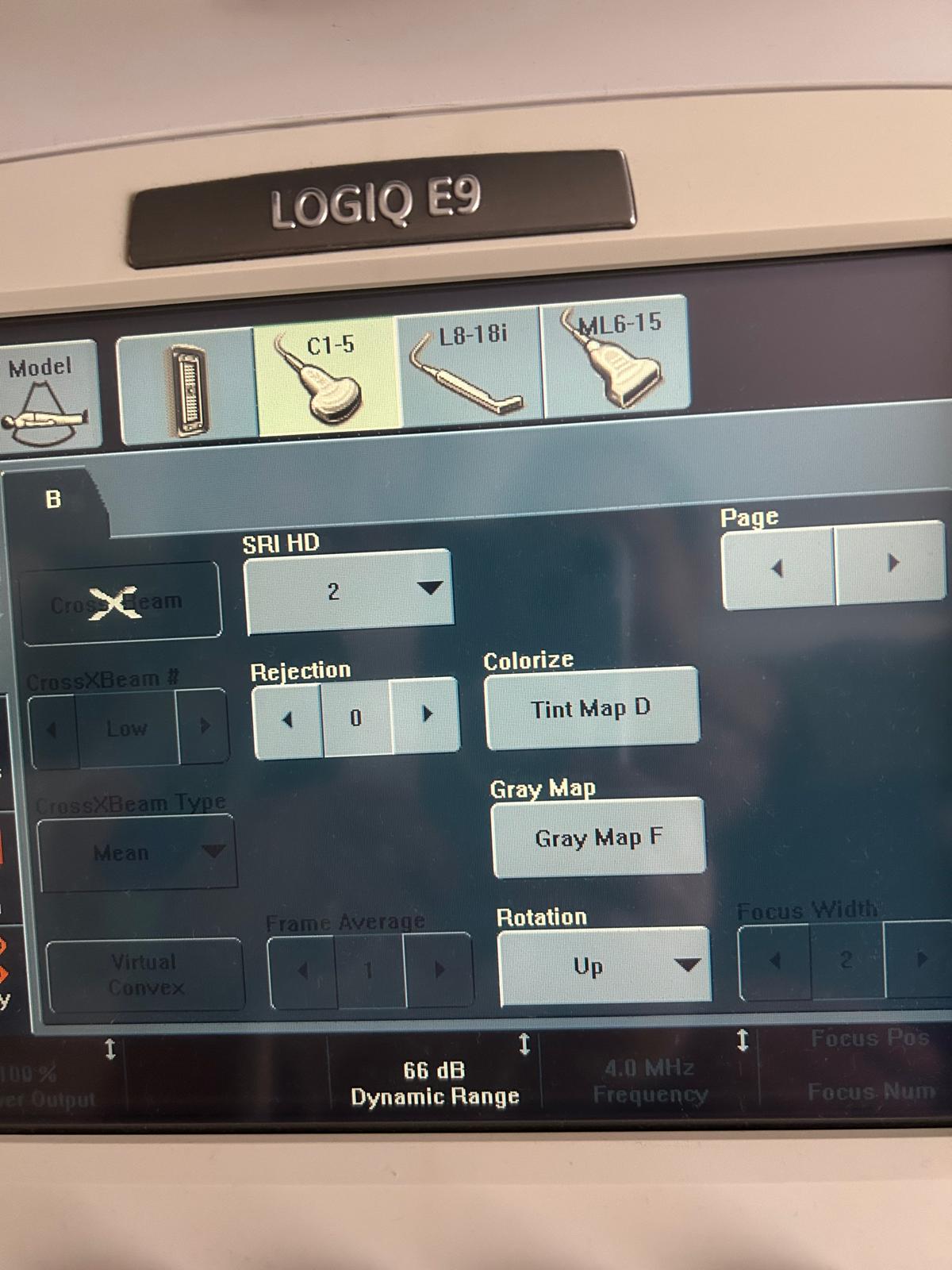
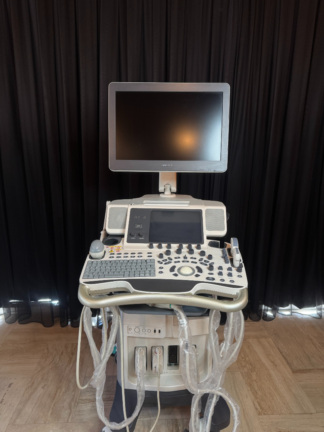
The probe models attached to the device and actively visible on the system screen (on the touch screen in the 3rd visual) are as follows:
- C1-5 Probe: Convex probe (For abdominal and OB/GYN use).
- L8-18i Probe: High-frequency “Hockey stick” type linear probe (For superficial, musculoskeletal, and small parts).
- ML6-15 Probe: Matrix Linear probe (For superficial imaging such as vascular, breast, and thyroid).
These probes are physically observed to be attached to the device’s probe ports. In total, 3 probes and 4 probe ports have been visually identified on the main unit. No significant visual breakage or crimping has been detected in the probe cables.
Screen and Control Panel Status
The device has a dual-screen structure. The main imaging monitor is at the top, and the touch command screen is at the bottom.
- Main Monitor: Operational, colors are vibrant, and pixel errors (dead pixels, lines) are not visible in the photos. The screen displays the date “25/03/25 15:40”. It is understood that the device’s system clock is set to March 2025.
- Touch Panel: The interface is clearly readable. No scratches or dullness are observed on the touch surface. Image enhancement functions such as SRI HD and CrossXBeam are seen to be active and controllable on the interface.
- Keyboard and Keypad: The QWERTY keyboard keys are complete. The TGC (Time Gain Compensation) slider keys (on the right side) are in place and generally centered, except for a few. No signs of heavy wear or fading are present on the trackball (cursor ball) and its surrounding keys.
General Physical Condition and Cosmetic State
Originality: The device’s labels, logo placements, software interface, and physical craftsmanship quality indicate that the product is an entirely original GE production.
Physical Deformations:
- No yellowing (very slight signs of yellowing), cracks, or fractures have been observed on the device’s outer casing (plastic parts).
- The gel bottle holders are clean.
- No impact marks have been found on the handles and edge barriers.
- There is no visible cable clutter at the bottom; the cables are tidy.
Cosmetic Comment: The device can be categorized as “Second Hand – Very Good” or “Well-Maintained Used”. Considering its 2011 manufacturing year, its condition is exceptionally clean for its age.
Potential Failure Risk and Current Failures
There is no evidence in the visuals that the device is displaying “Error” or “Warning” messages. The system has successfully powered on (“booted”) and entered imaging mode.
Note: Due to the software being from 2011 (BT11 version), processing speed may be slower compared to current technology, but this is not a malfunction. The connection points of the probe cables appear sturdy. No physical failure risk (e.g., rust, exposed cable) has been visually detected.
Quantity Information
The visual set subject to this report includes 1 GE LOGIQ E9 main unit and 3 integrated probes.