Ready For Sale
Secondhand Philips iU22 Ultrasound Machine
Price: USD$ 6.000,00 Approx: 270.000,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 20.11.2025
Philips Ultrasound Imaging System Detailed Analysis Report
Device Identification and Brand Detection
The brand and device information identified as a result of detailed examinations conducted on the medical imaging device in the visual are as follows:
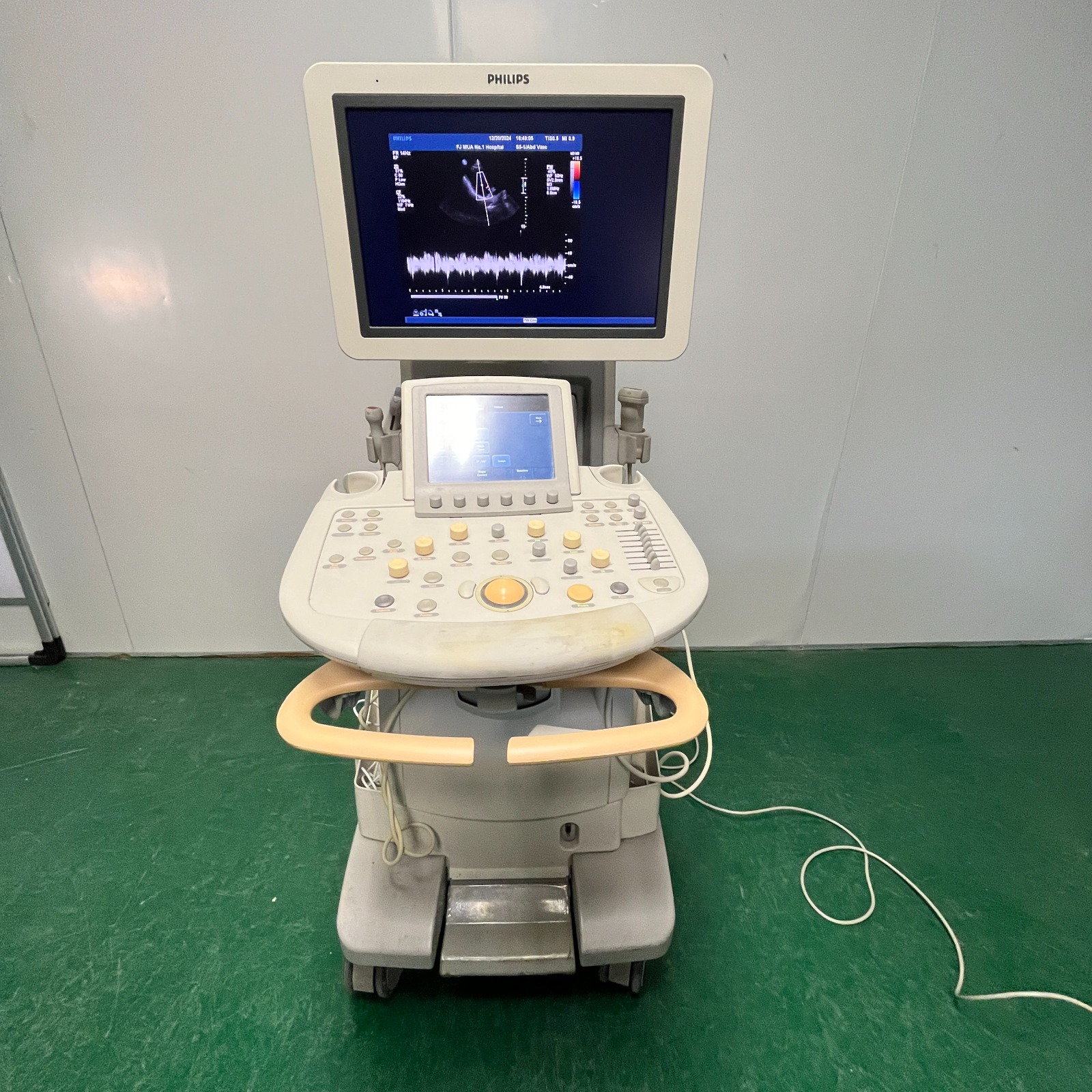
- Brand: A clear and legible PHILIPS logo is present on the top frame of the main screen of the device and in the upper left corner of the screen interface.
- Device Type: Considering the product’s form, control panel, probe holders, and the imaging data on its screen (Doppler waves), this device has been identified as an Ultrasonography (Ultrasound) System.
- Model Information: No specific model number (e.g., “Affiniti 70” or “EPIQ 5”) is visible in writing on the device casing or screen frame in the visual. Adhering to the principle of “no speculation,” the model number has not been specified.
- Institutional Affiliation: An institutional name, “FJ MUA No.1 Hospital,” was detected on the screen in the upper central part, indicating that the device was previously part of a hospital’s inventory.
Visual Condition and Cosmetic Deformation Analysis
The following details stand out when examining the general physical condition of the device:
- Control Panel Deformation: In the soft-textured area (palm rest) located directly below the device’s keyboard and control buttons, where the operator rests their wrist, there is severe discoloration, yellowing, and staining. This area has changed from its original color (likely light grey/white) to a yellow-brownish tone. This condition indicates intensive use.
- Casing Cleanliness: Superficial marks due to use are generally visible on the device’s lower body and plastic components, but no breaks or cracks have been detected.
- Handlebars: The “C”-shaped orange/salmon-colored handle bar at the front of the control panel is in place and robust. However, slight fading in the color tone is noticeable.
- Lower Assembly and Pedals: Wear marks due to use are present on the metal foot pedals located at the very bottom of the device. The wheel covers appear intact.
Technical Hardware and Screen Analysis
The device is operational, and the following technical details have been analyzed based on active screen data:
- Screen Condition: The device’s LCD screen is operational, and image clarity is at a good level in the visual. There is no visual evidence of dead pixels or screen cracks.
- Software Interface: An active ultrasound scanning session is visible on the screen. A blue/red Doppler flow window and a spectral waveform are present below it.
- Date and Time Information: The date 12/20/2024 and time 10:42:05 are displayed in the upper information bar of the screen. (This information indicates the date the photo was taken or the date the device’s system clock is set to).
- Technical Parameters: Ultrasound imaging parameters such as TIS0.8, MI 0.9, Fr 14Hz are readable on the screen. This data confirms that the device’s processor and software are currently active.
Control Panel and Mechanical Components
A detailed analysis of the control panel forming the user interface is as follows:
- Touchscreen: Below the main monitor, at the center of the control panel, there is a second small touchscreen interface, which is also illuminated, and menu items are visible.
- Physical Buttons and Knobs: Numerous rotary knobs and push-buttons are present on the panel. Although some buttons (especially those in the bottom row) show slight fading of their labels or colors, no physically missing buttons are visible.
- TGC Sliders: 8 TGC (Time Gain Compensation) adjustment sliders are fully present on the right side of the panel.
- Trackball: The yellow/orange trackball located in the center of the panel is fixed in place.
- Probe Holders: Vertical probe holder slots are present on the right and left sides of the panel. No mechanical breaks are observed in the slots.
Missing Parts and Accessories
The most prominent missing items in the visual are listed below:
- Ultrasound Probes: Despite the presence of probe socket inputs on the device and holders on the sides, no ultrasound probe (transducer) is attached in the visible area. The probe holders are empty.
- Cabling: Some white cables are seen hanging from the back or side of the device, but their ends are loose or irregularly positioned.
- Original Box: The product’s original box or packaging is not present in the visual.
Potential Malfunction and Risk Assessment
Risk analysis based on visual data:
No condition falling into the “Severe Damage” category, such as broken, shattered screen, or missing casing parts, has been observed on the device. There is no “System Error” or error message window on the screen; on the contrary, the normal measurement screen is active. This indicates that the electronic and software system is performing its basic functions. However, the extensive discoloration on the control panel suggests that the device may require hygienic cleaning or part replacement.
Quantity Information
The visual contains 1 unit of the Philips brand ultrasound device main body.