Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 24.10.2025
Siemens Acuson NX3 Elite Ultrasound Device Analysis Report
Overview and Device Identification
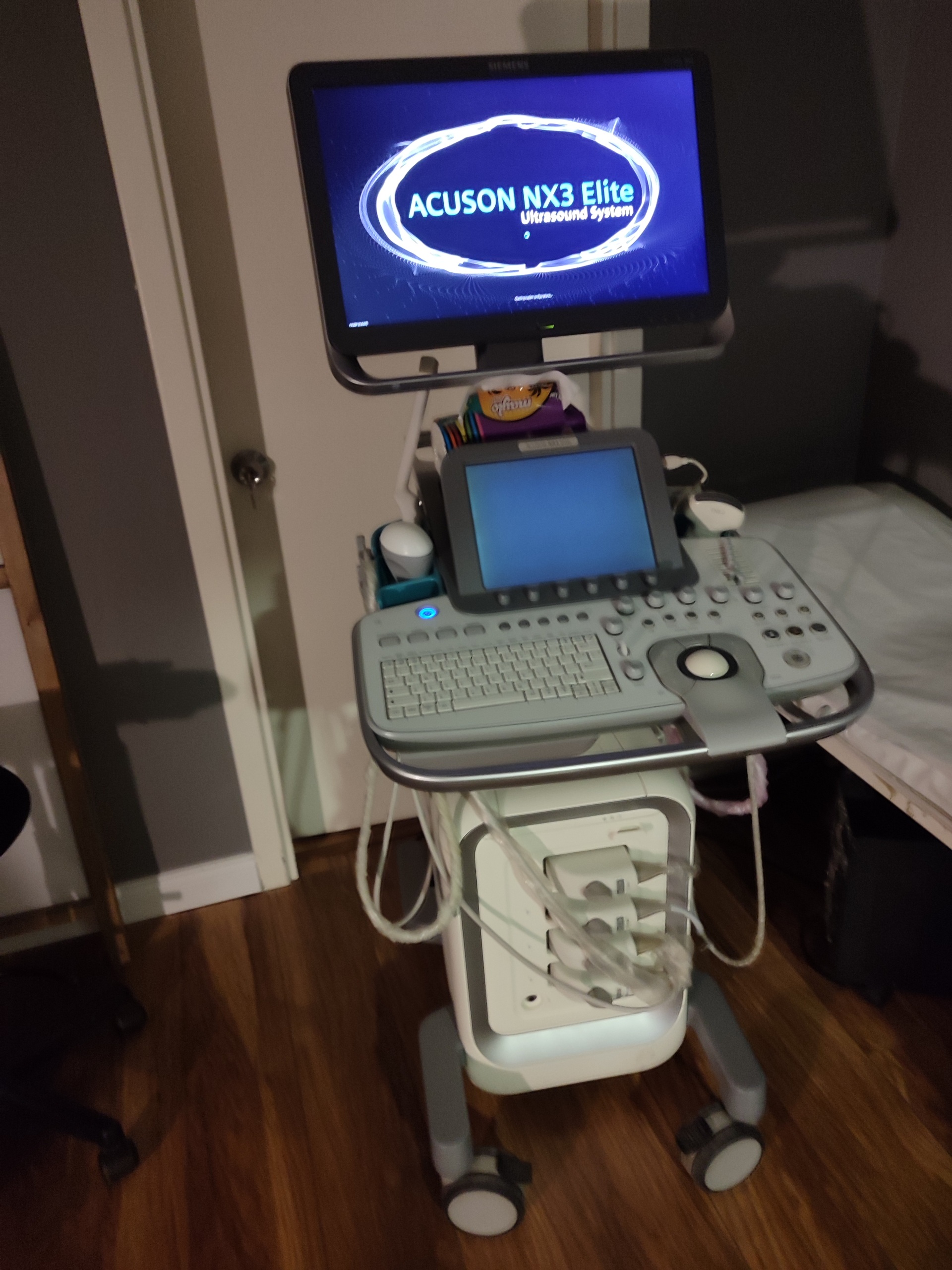
The device shown in the visuals is a wheeled ultrasound system used for medical diagnostic purposes. The device is observed to be powered on and its screens are active. It generally gives the impression of being lightly used and in a clean condition. The product consists of a main unit, a primary display monitor, a touch control panel screen, a keyboard, a trackball, and connected probes.
Brand and Model Information
As a result of a detailed visual inspection, the brand and model of the device have been clearly identified. On the startup screen of the main display, the phrase Siemens Acuson NX3 Elite Ultrasound System is clearly readable. This information 100% confirms the identity of the device. The device is a product used in the field of medical imaging.
Condition and State Assessment
- General Condition: The device is visually in very good and well-maintained condition. No significant wear or damage has been observed on it. This indicates that the device has been used carefully.
- Physical Deformations: There are no cracks, dents, or deep scratches on the white and gray casing of the device. The surfaces are clean, and no color fading is visible.
- Originality: The font of the “Siemens” and “Acuson NX3 Elite” inscriptions on the device, the logo placement, and the overall craftsmanship strongly support that the product is original.
Electronic and Mechanical Component Analysis
Upon visual inspection of the device’s electronic and mechanical components, the following details have been identified:
- Screens: The device has two screens. The main monitor is observed to be on and displaying the “Acuson NX3 Elite Ultrasound System” startup screen without any issues. The touch control panel screen is also illuminated but does not show an active menu; this could be a normal startup state. No scratches or pixel defects have been detected on either screen.
- Control Panel: The keyboard, function keys, and trackball are complete and in good physical condition. The blue power light on the control panel being on confirms that the device is receiving power.
- Mechanical Parts: The device’s wheels and wheel locking mechanisms are in place and appear functional. There are no breaks or deformations on the cable hangers and probe holders.
Accessories and Connection Ports
- Quantity Information: The visuals show 1 unit of the Siemens Acuson NX3 Elite ultrasound device.
- Probes (Transducers): Three ultrasound probes (transducers) are observed to be attached to the front panel of the device. The models or technical specifications of the probes cannot be determined from the visuals.
- Other Accessories: Next to the control panel, there is a white, mouse-like control accessory.
- Connection Ports: The sockets where the probes are connected appear clean and undamaged.
Potential Risk Assessment
As a result of visual inspections, there is no obvious evidence pointing to an existing defect in the device. The device’s general condition is very good, and it has been used cleanly. Only a white wrapping or tape on a cable bundle hanging from the side of the control panel draws attention; this situation might be an aesthetic flaw or serve a cable protection purpose, but its effect on current functionality cannot be understood from the visual. Other than this, it is assessed that the device has the potential for long-term use and does not carry a significant risk of malfunction.