Ready For Sale
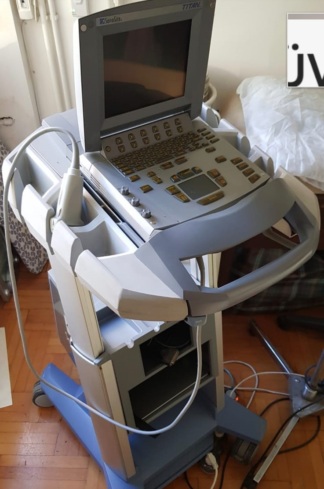
Secondhand Siemens Acuson Sequoia 512 Ultrasound Machine
Price: USD$ 1.375,00 Approx: 61.875,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 10.02.2026
ACUSON Sequoia 512 Ultrasound Imaging System Analysis Report
Device Detection and Identification
The examined visuals belong to a high-end ultrasound system equipped with a mobile stand, used for medical imaging. The device has a station structure consisting of an integrated CRT monitor, a mainframe, a control panel (keyboard, trackball, TGC sliders), and probe (transducer) ports. Additionally, specific ultrasound probes used with the system are also detailed in the visuals.
Brand and Model Information
Brand and model information obtained from a detailed analysis of the visuals is as follows:
- Main Device Model: The inscription located on the front of the device, just below the floppy disk drive, is clearly readable as ACUSON Sequoia 512.
- Monitor Brand: The SIEMENS logo is clearly visible on the frame of the display unit at the top of the system.
- Probe 1 (Linear): The third visual shows the inscription 8L5 acuson on the probe held by hand.
- Probe 2 (Convex/Sector): The fourth visual shows the inscription 6C2 acuson on the displayed probe.
Originality Status
Upon examining the print quality of the logos on the product, the industrial design of the casing, the panel layout, and the label fonts, the product is assessed as completely original. The device bears characteristic design features from the period of or after the Siemens and Acuson merger.
Areas of Use
This system is a professional medical diagnostic device designed for hospitals and clinics. It is used for imaging internal body structures in fields such as Radiology, Cardiology, Obstetrics/Gynecology (OB/GYN), and Vascular Surgery.
- General System: Used for color Doppler imaging and detailed ultrasonography procedures.
- ACUSON 8L5 Probe: Typically a linear probe operating in the 8 MHz frequency band. Suitable for superficial tissues, vascular imaging, thyroid, breast, and musculoskeletal system examinations.
- ACUSON 6C2 Probe: Typically a curved probe, used for abdominal organs, obstetric, and gynecological examinations, operating at low-to-medium frequencies.
Quantity Information
The visuals show a total of 1 ultrasound main console (including monitor and stand) and 2 external ultrasound probes (8L5 and 6C2). The main unit has multiple probe ports.
General Condition and Physical State
The device is in “used” condition based on its overall appearance. There are cosmetic indications that may be attributed to long-term use and exposure to humidity/heat.
Physical Deformation Examination
The product shows visually distinct signs of aging that do not affect functionality:
- Color Change: Yellowing (ABS plastic aging) is observed on the plastic parts of the device, particularly around the main casing and the floppy disk drive, occurring over time.
- Surface: Slight scuff marks are observed around the monitor casing and control panel. The base (feet) of the device may have dirt and minor deformations due to use and impacts.
- Label Residue: In the upper left corner of the main unit, a blue-colored residue resembling an old service or calibration sticker and wear are visible.
Mechanical Components
The device’s stand, wheeled structure, and foot pedals (brake mechanism) are present in the visuals. The control panel, keyboard keys, TGC (Time Gain Compensation) sliders, rotary knobs, and trackball (cursor ball) appear physically complete. No fading or missing keys were detected in the visuals.
Electronic Components and Screen Analysis
The device is in the off position in the visuals, therefore no comments can be made regarding screen performance (pixel errors, image quality). The monitor is an old-type display with CRT (cathode ray tube) technology. While the electronic condition of the keypads on the control panel appears physically sound, their functionality should be tested.
Accessories
The most important accessories displayed on the device and subject of this report are:
- 1 Unit ACUSON 8L5 Ultrasound Probe
- 1 Unit ACUSON 6C2 Ultrasound Probe
Cable hangers and probe holder slots are present on the front of the main unit. The device’s power cables are visible extending from the bottom, but their complete integrity cannot be confirmed from the visuals.
Labels and Technical Specifications
Readable technical details from the visuals are as follows:
- Main Brand: ACUSON Sequoia 512
- Monitor: SIEMENS
- Probe Codes: 8L5 and 6C2
Since the rear panel label is not visible, the Serial Number, Place of Manufacture, and Electrical Values (Volt/Hz) cannot be read.
Size and Compatibility
The product is a large-sized (mobile cart type) device, designed to be positioned in a room within a hospital environment, and is adult-sized. It is not a portable laptop-type model.
Estimated Year of Manufacture
The production year label is not visible on the product. However, the device having a CRT monitor, the market history of the “ACUSON Sequoia 512” model, and the presence of a floppy disk drive indicate that the device was likely manufactured between the late 1990s and early 2000s. This is a vintage medical technology product.
Existing Faults and Risk Analysis
The visuals do not show any “definite fault” indicators such as a broken, cracked screen, or severed cables. However, potential risks are as follows:
- CRT Lifespan: Cathode ray tube monitors have a risk of losing their brightness over time.
- Data Storage: Floppy disk drive technology is obsolete today; data transfer might be challenging.
- Probe Membranes: It is not 100% clear from the visuals whether there is wear on the patient-contacting surfaces (membranes) of the probes, but they appear very clean.
General Assessment
ACUSON Sequoia 512 is one of the top-segment (high-end) devices of its era. The visuals show that the device’s casing maintains its integrity, the keypad appears complete, and it includes two original probes. Cosmetic yellowing is normal for the device’s age. It is in a condition that can be considered for medical spare parts, educational use, or clinical use after refurbishment.