Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 08.10.2025
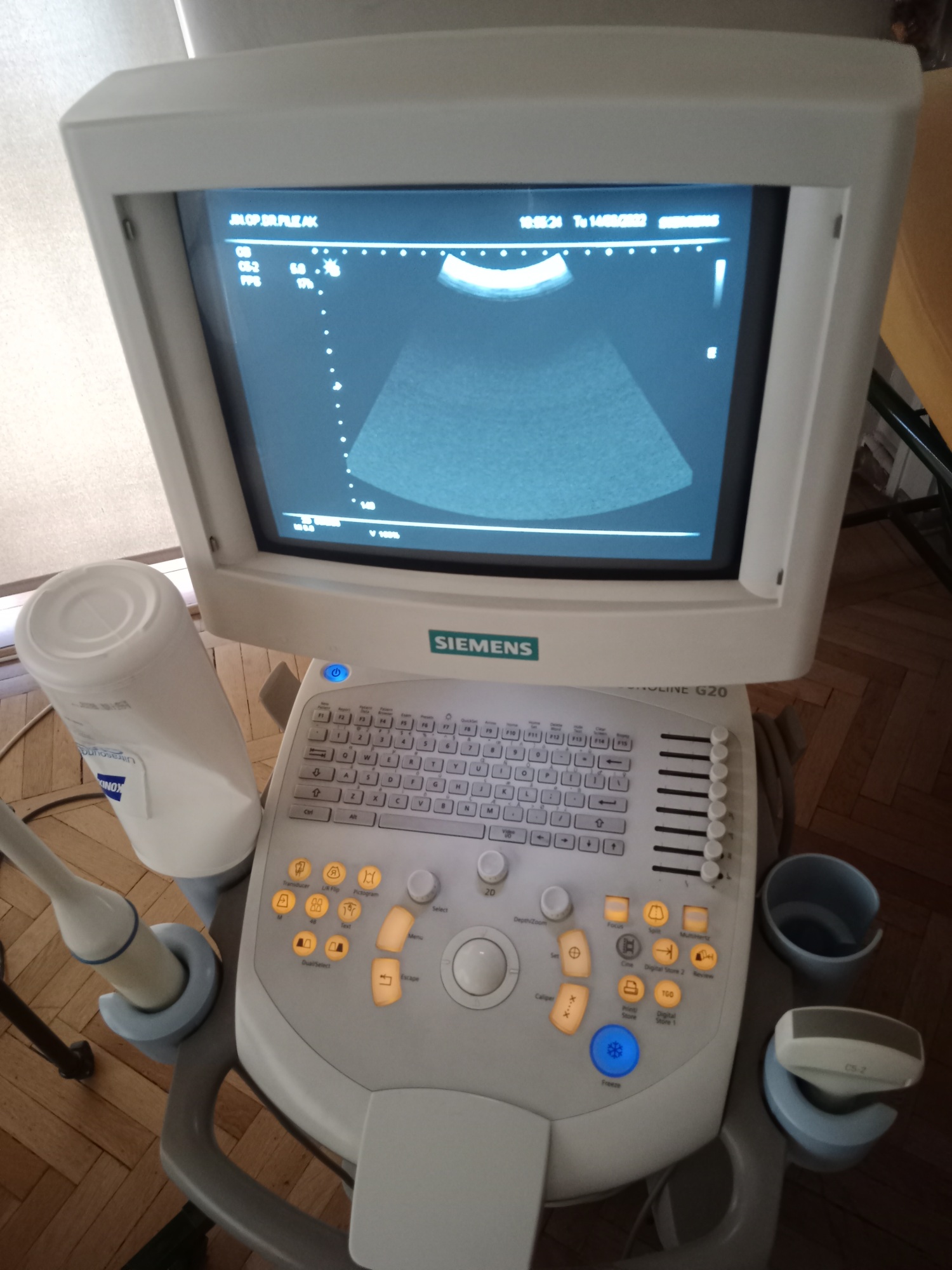
Siemens Sonoline G20 Ultrasound Device Analysis Report
Product Identification and Overview
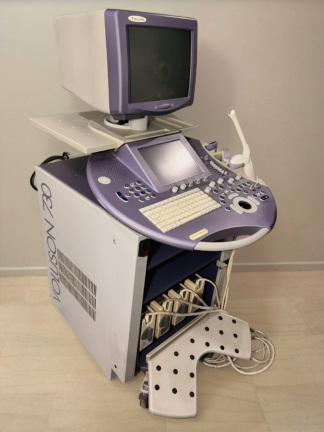
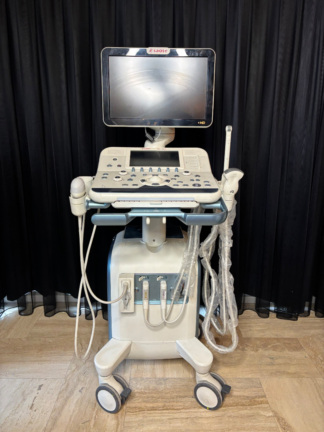
The images display an ultrasound device and its accessories, mounted on a wheeled stand, used for medical imaging purposes. The device is designed for diagnostic ultrasonographic examinations in clinical settings. The overall structure of the system consists of a main unit, monitor, control panel, integrated printer, and ultrasound probes. The general design of the device is compact and portable, allowing for easy maneuverability in confined spaces.
Brand and Model Information
The brand and model information of the devices shown in the images are detailed below. This information has been clearly identified from the labels and inscriptions on the devices.
- Ultrasound Device Brand-Model: Siemens Sonoline G20
- Ultrasound Probe Model: C5-2
- Video Printer Brand-Model: Mitsubishi P93
- Ultrasound Gel Brand: Konix
General Condition and State
The device is generally a used product. When powered on, the screen and control panel button backlights are observed to be working, indicating that the device’s basic electronic functions are active. A slight yellowing is observed in the plastic components of the device, attributable to age and use. However, its general condition does not appear to include significant damage that would impede its functionality.
Physical Assessment
Upon detailed examination of the device’s outer casing, no significant cracks, breaks, or dents were detected. Only minimal scratches and color changes, resulting from use and time, are present. Plastic parts such as the device’s wheels and probe holders are in place and appear physically sound.
Electronic and Mechanical Component Analysis
The electronic components are understood to be operational from the images of the device in the on state. The buttons on the control panel and the rotary control knob appear to be physically in place. The integrated Mitsubishi P93 model printer’s display shows an “EP” warning. This warning usually stands for “End of Paper” or “Empty Paper,” indicating that there is no thermal paper in the printer or it has run out. Mechanically, the sockets where probes are attached and other movable parts are visually in good condition.
Accessories and Equipment
The accessories supplied with the device in the images are as follows:
- Quantity Information: A total of 1 ultrasound main unit, 2 ultrasound probes, 1 integrated printer, and 1 bottle of ultrasound gel are present.
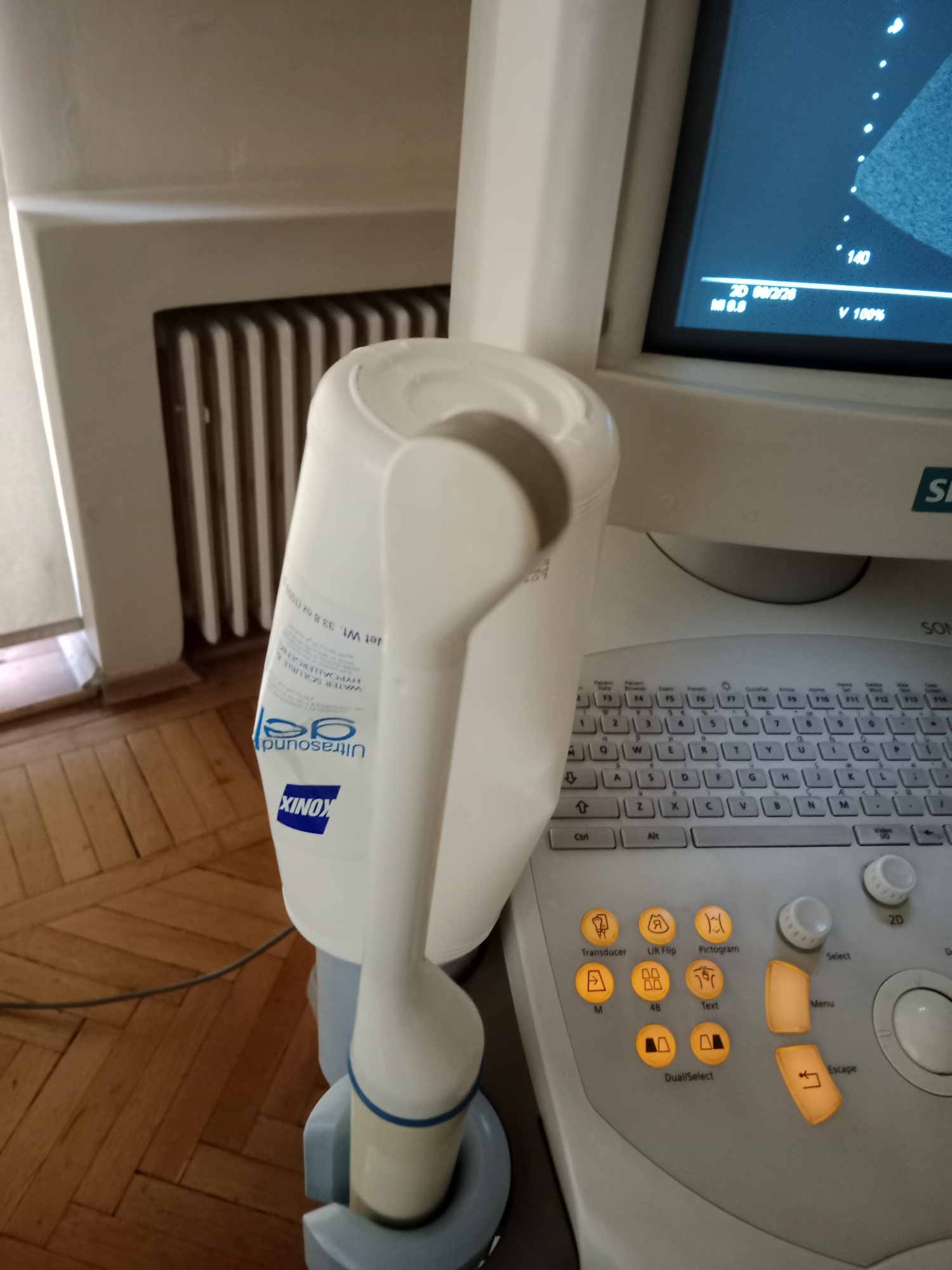
- Ultrasound Probes: One convex probe with “C5-2” written on it and one longer, thinner probe of an unreadable model, likely an endocavitary/vaginal type, are available. No visible damage has been detected on the cables and connectors of the probes. The C5-2 probe is generally used for abdominal, obstetric, and gynecological imaging.
- Printer: One Mitsubishi P93 model medical thermal video printer is integrated into the device.
- Ultrasound Gel: One bottle of Konix brand ultrasound gel, understood to be 1000 ml / 33.8 oz, is located in the probe holder. The bottle appears to have been used.
Screen Analysis
The device’s screen is an old-type CRT monitor. The screen is operational and displays an ultrasound image along with various technical parameters (G, DR, D, FPS, MI, TIS) and date/time information (“19:55:24 Tu 14-OCT-2025”). In the upper left corner, there is text “UL.GP.SKR.PLECAK”; this text is likely a patient record, department, or clinic designation.
Potential Risks and Points to Consider
Although the device’s general condition indicates it is in working order, there are some points for the buyer to consider. The yellowing of the device’s plastic casing points to long-term use and the age of the product. The integrated printer displaying an “EP” warning indicates that thermal paper replenishment is required for its operation; this is not a malfunction but a consumable material deficiency. Generally, no serious wear or damage suggesting an immediate risk of failure has been observed in the device.