Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 08.12.2025
QV-500 Portable Vein Finder Analysis Report
Device Identification
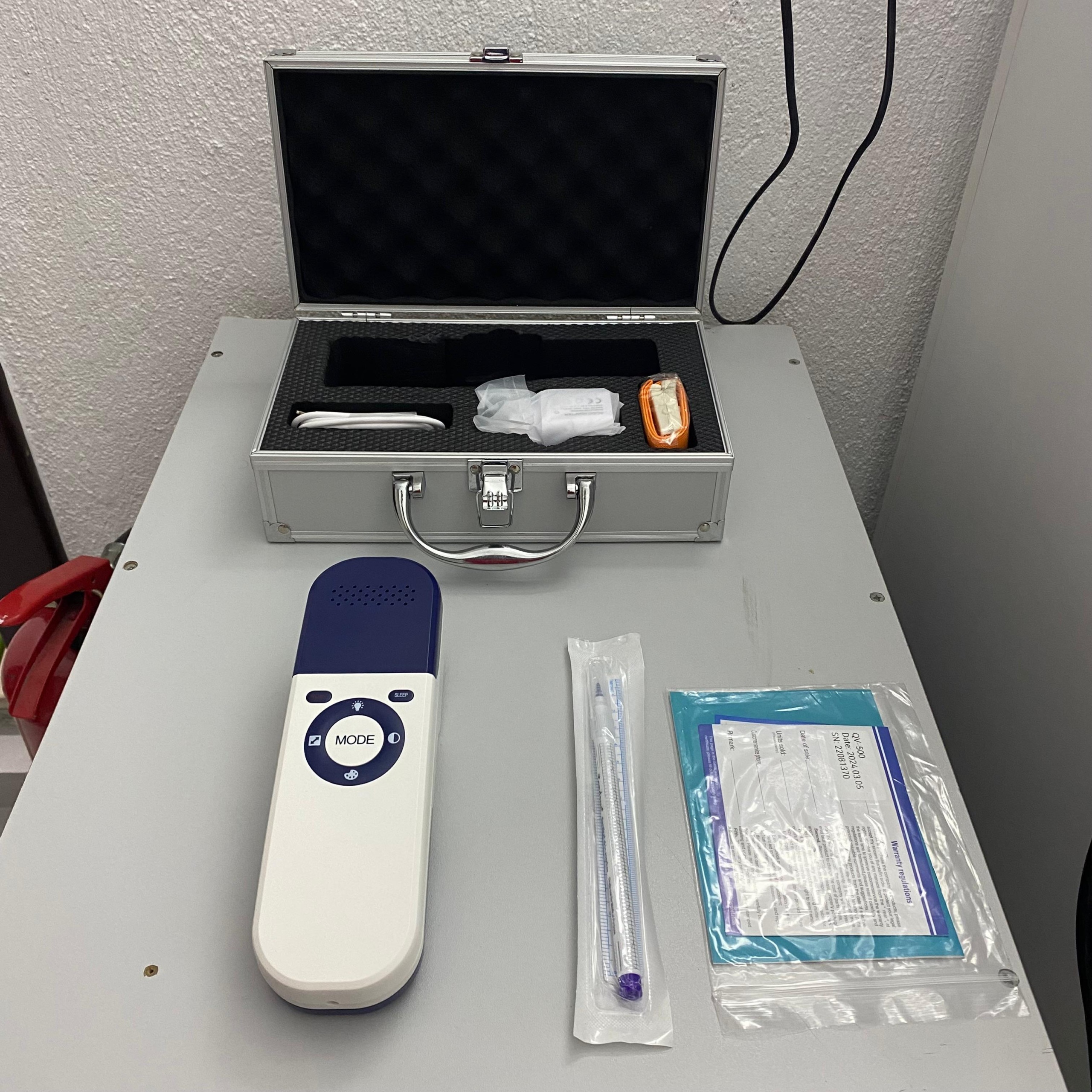
The examined visuals show a portable vein finder used in medical procedures and its carrying set. The product is a handheld electronic device designed to help healthcare professionals see veins more clearly. The set consists of the device itself, a carrying case, charging equipment, and a marking pen.
Brand and Model Information
Based on the clear indications on the document and warranty card in the visuals, the device model has been identified as QV-500. The brand of the surgical skin pen provided with the device is clearly legible as Tondaus in the visual. The “QV-500” inscription is clearly visible as the model number in the upper right corner of the warranty/user manual document.
Originality and General Condition
The product’s casing, keypad structure, accompanying aluminum carrying case, and internal foam cutouts reflect an industrial standard of manufacturing quality. The fact that the equipment is packaged and the labels are neatly positioned indicates that the product is an original set. The cosmetic condition of the device is “New” or “Lightly Used/Display”. No signs of wear due to use have been found.
Quantity Information
A total of 6 main parts have been identified in the set:
- 1 pc QV-500 Vein Finder Device
- 1 pc Aluminum Carrying Case (With locking mechanism)
- 1 pc Charging Adapter (In plastic wrap)
- 1 pc USB Connection/Charging Cable
- 1 pc Tondaus brand Surgical Skin Marker (In its packaging)
- 1 pc Carrying Strap/Lanyard (Orange colored, packaged)
Physical Deformation and Cosmetic Examination
No cracks, scratches, dents, or discoloration are observed on the device’s outer casing, screen panel, or control buttons. The joints of the blue and white plastic casing are smooth. There is no peeling or deformation on the dark blue rubber/plastic coating of the grip handle. The product is visually in perfect condition.
Mechanical and Electronic Components
The front face of the device features a circular control panel. This panel includes the main “MODE” button, “SLEEP” button, and peripheral control icons. There is no fading on the buttons. The upper part of the device has a perforated structure, presumed to be ventilation grilles or a speaker output. The carrying case’s hinges, carrying handle, and metal locking mechanism appear sturdy, with no signs of corrosion or rust.
Keypad and Function Analysis
Upon examining the control panel in the visual, the following functional buttons are observed:
- Center: The large “MODE” button, which changes the device mode.
- Top: A light bulb icon symbolizing illumination/brightness adjustment.
- Bottom: A palette icon with the function to change color/image tone.
- Left: Diagonal arrows indicating image size or zoom.
- Right: A crescent moon icon indicating contrast or day/night mode.
- Side: The “SLEEP” button for power saving or standby mode.
Label and Document Analysis
The visual shows a blue-white colored document within a transparent folder. The legible information on this document is as follows:
- Model: QV-500
- Date: 2024.03.05 (This date could be the production or sales date, indicating the product’s recency).
- SN: 22081370
- Document Title: Warranty regulations.
Condition of Accessories
The charging adapter and cable are in their transparent bags with their factory windings intact. This suggests that the accessories have never been used or have been carefully repackaged. The accompanying “Tondaus Surgical Skin Marker” is in its sterile packaging and has not been opened. The aluminum case, with its black protective foam inside, is structured to protect the device from impacts.
Areas of Use
This device is used to detect the location of subcutaneous veins using infrared or similar technology and project them onto the skin surface. It is designed to facilitate blood collection and cannula insertion, especially in infants, the elderly, obese patients, or patients with difficult-to-find veins. It is suitable for use in hospitals, clinics, blood collection centers, and emergency departments.
Potential Failure Risk
As a result of the visual inspection, no signs of physical damage, wear, or misuse have been detected on the product. The integrity of the cables and the “new” appearance of the device indicate a low risk of physically-sourced failure in the short term.