Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 18.05.2026
Report code: 1779087667
Gait Trainer Rehabilitation Device Analysis Report
Device Identification
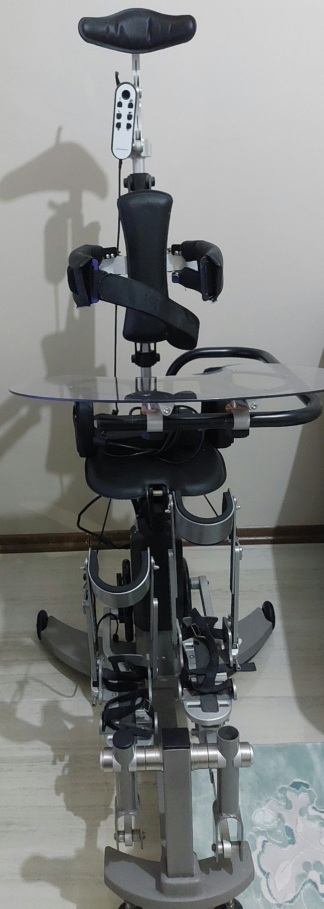
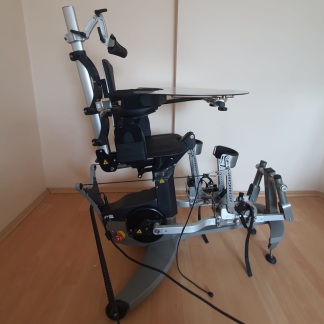
The product in the image is a gait trainer device used in physical therapy and rehabilitation processes, featuring a wheeled chassis that supports the patient in an upright position and allows for walking exercises.
Brand and Model
No readable brand or model information could be identified on the device’s body, labels, or packaging.
Usage Areas
This device is used in rehabilitation centers and hospital physical therapy departments to help patients with restricted walking ability due to neurological or orthopedic conditions regain walking skills, increase muscle strength, improve balance, and support the cardiovascular system. It provides a safe walking experience by supporting the patient from the torso, hip, and leg regions.
Quantity Information
The image shows 1 main device and various support apparatuses integrated into the device (headrest, torso supports, front bar with tray, handles, safety belts).
General Condition
The general condition of the device appears to be good. No significant wear or damage is visible on the metal and plastic components. However, there are non-original materials (packing tape, cardboard box, plastic bag) on and around the device.
Physical Deformation
No serious physical deformation such as deep scratches, dents, or cracks has been observed on the white painted chassis and metal parts of the device. The black handle sponges and upholstered parts appear intact. It is observed that packing tape is wrapped around the end of the left handle.
Mechanical Components
The device’s wheels, height adjustment mechanisms, joints of the support arms, and connection points appear visually intact and complete. The adjustment knobs are in place. A cardboard box is wedged in the middle section of the device, under the seat or support section. Additionally, there is a transparent plastic bag tied onto the chassis at the rear.
Electronic Components
There is a small screen panel on the front of the device, between the handles. The status of this panel cannot be clearly understood from the image. In the lower rear part of the device, there is a black section thought to be the motor or battery block, and loose, disorganized cables and sockets protruding from this section are visible. Some of these cables are exposed and positioned in a disorderly manner.
Accessories
The device features a headrest, belts and pads that secure the torso, a support bar with a transparent tray at the front, and handles. A black part, which could be a remote control or a connection apparatus, is hanging within the cable clutter at the rear. Socket inputs or connection terminals can be seen at various points on the device’s chassis.
Battery Status
The black block at the lower rear of the device likely houses the battery and motor unit. No comment can be made regarding the battery’s capacity, charge status, or physical condition (swelling, leakage, etc.) based on the image.
Label Information
There are several different labels on the device’s chassis. A label containing technical information is visible on the black unit at the lower rear, warning or instruction labels are on the side of the chassis, and a barcode label is visible near the front wheel. However, specific information such as serial numbers, lot numbers, or REF codes on these labels cannot be read due to image resolution.
Screen Analysis
The small screen on the front of the device is turned off. There is no text, warning, or menu display on the screen.
Size and Compatibility
The adjustable structure of the device indicates that it can accommodate patients of different heights and weights. However, information regarding a specific user size (child/adult) or weight capacity is not available in the image.
Existing Faults
No clear evidence of a fault, such as a broken part or an error message on the screen, is visible on the device. However, the fact that the cables at the rear are disorganized and exposed, packing tape is wrapped around a handle, and a cardboard box is wedged into the device indicates irregular usage.
Potential Fault Risk
The fact that the electronic connection cables and sockets at the rear of the device are disorganized, exposed, and unprotected poses a serious risk of failure that could lead to these parts getting snagged, severed, or causing a short circuit. This situation may jeopardize the reliability of the device’s electronic systems. Although the mechanical structure of the device appears generally sound, this disorder in the electronic components is a potential risk factor.