Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 09.05.2026
Report code: 1778316032
Achilles Ultrasonic Bone Densitometer Analysis Report
Device Identification
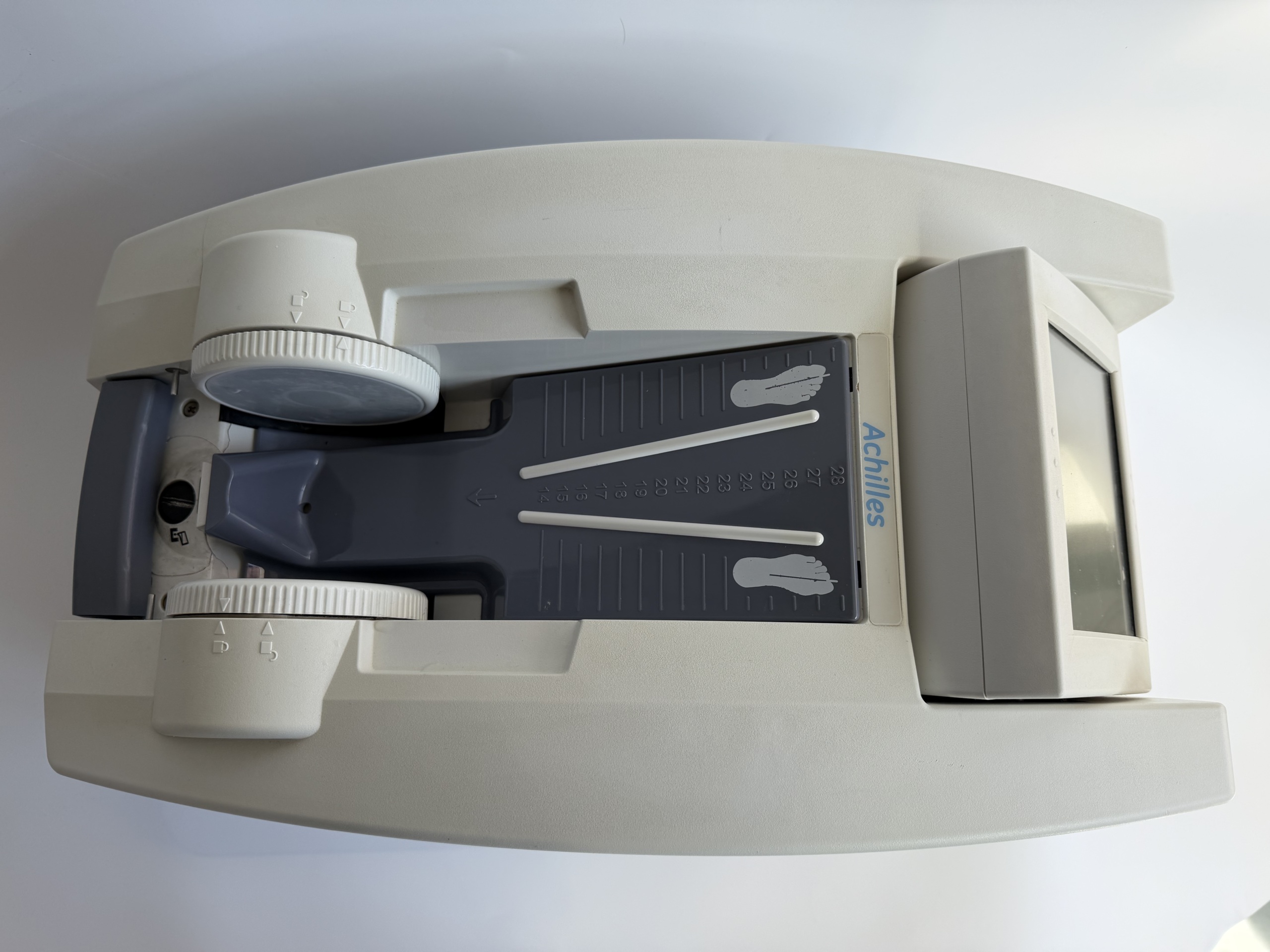
The device in the images is an ultrasonic bone densitometer used for measuring bone density from the heel.
Brand and Model
The Achilles brand is clearly legible on the device. Model information could not be identified in the images.
General Condition and Physical Deformation
The outer casing of the device consists of light gray and dark gray plastic parts. Slight discoloration and superficial scratches due to usage are visible on the plastic surfaces. The general condition of the device is used. No fractures or cracks have been detected on the casing.
Mechanical Components
On the upper part of the device, there is a dark gray section designed for heel placement, featuring footprint graphics and a scale numbered from 14 to 28. On both sides of this section, there are white adjustment wheels with lock/unlock symbols. The mechanical status of these wheels cannot be determined from the image. At the rear of the device, there is a display unit positioned at an upward angle.
Electronic Components and Display
There is an integrated display at the rear of the device. The display is off in the images; therefore, no information regarding its functionality or screen integrity could be obtained. The internal electronic components or socket inputs of the device are not visible in the images.
Accessories and Battery
There are no power cables, connection cables, gel, or other accessories belonging to the device in the images. There is no visual evidence regarding whether the device has an internal battery or its external battery status.
Label Information
No technical labels or barcodes indicating a serial number, lot number, REF code, or manufacturing year are visible on the device.
Size and Compatibility
The foot scale on the device ranges from 14 to 28. This scale indicates that the device is suitable for adult patients with different foot sizes.
Existing Faults and Potential Risks
There is no clear physical evidence in the images, such as broken parts, to suggest that the device is faulty. The outer casing of the device maintains its integrity. Since the screen is off, it could not be determined whether there is an electrical fault. Based on its visual condition, no significant wear or corrosion is observed, and the physical state of the device appears to be good.