Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 25.04.2026
Report code: 1777127187
Medical Aesthetic Laser Device Analysis Report
Device Identification
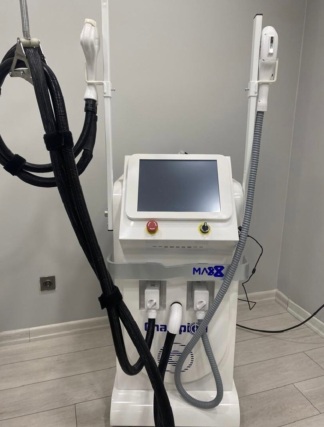
The images show a desktop-type medical aesthetic laser device main unit and its accompanying accessories. The device has a blue and gray plastic casing.
Brand and Model
No brand or model name can be clearly read on the main unit of the device. Therefore, brand and model information is not specified.
Usage Areas
The design of the device, the handpiece, and the accompanying ‘Light Guide’ accessories indicate that the product is a laser or intense pulsed light (IPL) system used in dermatology and aesthetic fields, likely for hair removal or skin treatments. The exact intended use cannot be determined without technical documentation.
Quantity Information
The following parts have been identified in the images:
- 1 laser device main unit
- 1 handpiece (probe) connected to the device
- 1 foot pedal
- 2 ‘Light Guide’ crystals in a transparent plastic box
- 1 empty cardboard box (labeled M100 Lens-unit)
General Condition and Physical Deformation
The outer casing of the device main unit maintains its integrity. No significant breaks or deep cracks are visible on the gray upper part or the blue lower part. However, there are friction marks, scratches, and discoloration on the outer surface of the handpiece (probe), particularly on the tip and body. The handpiece cable is wrapped with a protective covering. Signs of use and dust are also visible on the foot pedal. The transparent accessory box and the crystals inside appear clean.
Mechanical and Electronic Components
The front panel of the device features an LCD screen, a red emergency stop button, a key switch, and a rotary control knob. These parts are in place and appear intact from an external view. There is a socket input on the bottom right corner of the device where the handpiece is connected. Since the rear panel or internal components are not visible, no comments can be made regarding its electronic status. Whether the device is in working condition cannot be determined from the images.
Accessories and Label Information
The accessories provided with the device are as follows:
- Foot Pedal: A gray, wired foot pedal is present.
- Handpiece: There is a gray handpiece connected to the device with two white buttons on it.
- Light Guide Crystals: There are two crystal blocks inside a transparent plastic box. The label on the box contains the BIOS Medical Aesthetic Devices logo, the phrase Light Guide, and λ 620nm (wavelength) information. Additionally, the web address www.biosgroup.eu is written.
- Cardboard Box: An empty box is visible with the the SCOPE logo, USB Microscope M2, M100 Lens-unit (x100) text, and a barcode (4 571109 431041). The direct relationship of this box to the laser device cannot be fully understood from the images.
There is no technical label on the device main unit showing a serial number, model number, or year of manufacture in the images.
Screen Analysis
The LCD screen on the front panel of the device is off. There is no image, text, or error message on the screen.
Documents and Usage Duration
No documents such as invoices, warranty certificates, or user manuals belonging to the device are visible in the images. No information regarding the usage hours or duration of the device could be obtained.
Existing Faults and Potential Risk of Failure
No clear mechanical breakage or damage that would prevent the device from operating has been observed in the images. However, the significant wear and scratches on the handpiece (probe) indicate that this part has been used extensively. The fact that the handpiece cable is wrapped suggests that the cable integrity should be checked. The general condition of the device is used. Whether the device works electronically cannot be known without testing.