Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 18.12.2025
VORTEX Multi Epilation Device Analysis Report
Device Identification
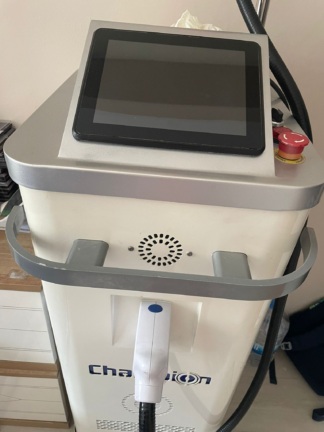
The product analyzed in the image is an upright type aesthetic procedure device used in professional aesthetic and beauty salons. The device has been identified as an epilation device (likely using Diode Laser or IPL technology) based on its body structure, cooling channels, user interface, and integrated handpiece systems. The main unit has a wheeled (cabinet) structure and features a control panel and handpiece holders.
Brand and Model
Upon examining the visual data on the product, the device’s brand and model can be clearly read:
- Brand: The inscription VORTEX is clearly visible on the device’s top panel and in the logo below the control screen.
- Model: The term Multi is located directly next to the brand name. In light of this data, the device’s brand and model have been identified as VORTEX Multi.
Authenticity
Upon examining the device’s metal craftsmanship, paint quality, the symmetry of its logo (the special red and black design of the “V” logo), and the font quality of the brand model inscription, the product is assessed to be an original VORTEX production. No incompatible parts or poor-quality labeling suggesting it is an aftermarket or assembled product have been observed.
Areas of Use
The VORTEX Multi is designed for use in aesthetic centers, beauty salons, and dermatology clinics. Its primary purpose is the permanent removal of unwanted hair (Laser Epilation). It is understood that the device is a medical/aesthetic equipment requiring professional use and is not suitable for home use.
General Condition and Physical State
The device’s general condition can be described as “used but clean”. No deep scratches, noticeable dents, or corrosion marks are visible on the cream/white paint of the metal body. The device’s external cosmetic state indicates careful use. From the floor tiles and wall reflection, it is understood that the device is in an indoor setting, likely a clinic.
Quantity Information
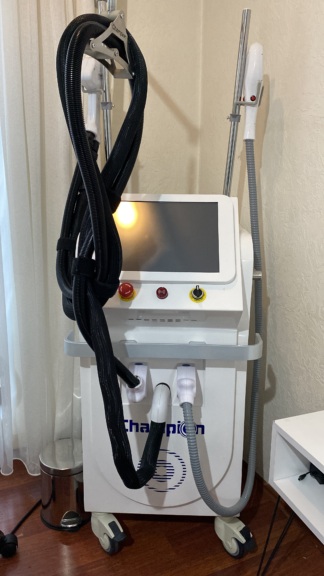
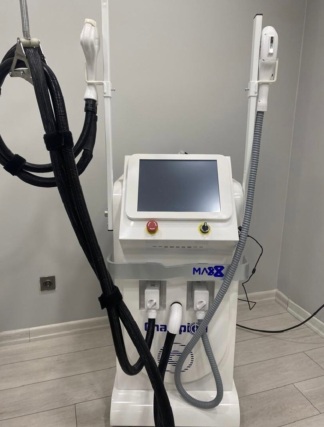
The image shows 1 (one) main device unit and 2 (two) treatment handpieces integrated with this device.
Mechanical and Control Components
The mechanical parts and control elements on the device have been analyzed as follows:
- Emergency Stop Button: A red, mushroom-headed emergency stop button is located on the panel. The button is intact and in place.
- Key Switch: On the right side, there is a key slot used to protect the device from unauthorized use or to turn it on/off. Although it is not clearly visible whether the key is inserted due to the viewing angle, the slot appears undamaged.
- Body and Ventilation: Ventilation holes (grilles) are present on the side parts of the top surface. These areas are open and not blocked, designed for the cooling system’s air intake.
Electronic Components and Screen Analysis
At the center of the device is a large, likely 10-12 inch, black-framed touchscreen panel. Since the device is currently off, the screen is dark, but no significant cracks or breaks have been detected on the screen surface. The reflective quality of the screen indicates a smooth surface.
Accessories and Cable Condition
The device’s most critical components, the treatment handpieces and cabling system, have been examined in detail:
- Treatment Handpieces: Two white-colored treatment handpieces are hanging in their holders on the front of the device. The handpieces have blue circular details on them. No breaks or cracks are visible in the plastic components of the handpieces.
- Hoses and Cables: The black spiral-protected hoses leading to the handpieces and the thick white/gray spiral main hose coming from the right side of the device appear intact. No deformations such as bending or tearing at the points where the black hoses connect with the handpieces have been detected in the image. The cable housings are in good physical condition.
Potential Malfunction Risk
As a result of the visual inspection, no obvious damage (break, severed cable, dented panel) that would impede operation has been observed on the device’s external hardware, cables, or screen. The cosmetic condition of the device is good, which indicates a low risk of mechanical malfunction. However, since the device is turned off, no comments can be made from the image regarding its internal electronic condition, software functions, or laser firing performance.