Ready For Sale
Secondhand AMS 200 Anesthesia Machine
Price: USD$ 3.000,00 Approx: 135.000,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 10.04.2026
Report code: 1775823690
AMS 200 Anesthesia Machine Analysis Report
Device Identification and Brand Model Information
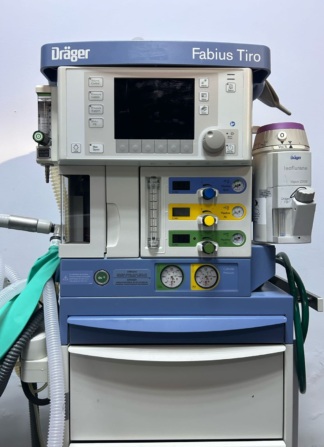
The product in the image is an anesthesia machine used in operating rooms. The inscription AMS 200 ANAESTHESIA WORKSTATION is clearly legible on the front panel of the device’s main body. The brand and model information AMS Blease 8200s is located on the ventilator module positioned at the very top of the device. Additionally, the AMS logo is present on the wheels at the bottom of the device.
Usage Areas
This device is used in operating rooms to deliver anesthetic gases and oxygen mixtures to patients during surgical operations, as well as to mechanically control the patient’s respiration. Through its integrated equipment, it provides gas flow to the patient via the breathing circuit and manages vital respiratory parameters.
Quantity Information
The image shows 1 main anesthesia workstation, 1 ventilator module integrated into the device, 1 vaporizer, and 1 set of breathing circuits (hoses and reservoir bag) connected to the device.
General Status and Condition
The device is in a used condition and appears to be an older generation model. There are superficial abrasions due to use on the body, slight discoloration on the plastic parts, and signs of wear on the stainless steel work surface. The device generally maintains its integrity but shows signs of wear and tear.
Physical and Mechanical Component Analysis
No major breaks, cracks, or dents have been detected on the outer casing of the device. However, there is a notable condition regarding its mechanical integrity:
- Three wooden blocks have been placed under the triple white drawer unit located at the bottom of the device to support the drawers from below. This indicates a mechanical collapse or failure in the drawer rail system or the supporting chassis.
- There are four wheels supporting the device. The surfaces of the wheels show dirt and wear.
- The transparent carbon dioxide (CO2) absorber canister on the left side is empty and appears physically intact.
- A vaporizer mounted on the right side of the device is mechanically fixed in place.
Electronic Components and Screen Status
The AMS Blease 8200s ventilator module at the top features an analog pressure gauge and digital display panels. Since the device is in the off position, no data or lights are visible on the digital screens. Control knobs, rotary potentiometers, and keypads are physically in place; no missing or broken buttons were detected.
Accessories and Connection Points
The device includes the essential breathing circuits and connections required for anesthesia administration:
- White, corrugated patient breathing hoses are lying scattered on the work surface.
- A green anesthesia reservoir bag is located among the breathing circuits.
- Black thick spiral cables and thin yellow gas hoses are connected to the valve and canister module on the left side of the device.
- A large medical gas cylinder fixed to the device is visible at the rear right side.
Label and Indicator Information
The following inscriptions and indicators have been identified on the front panel and modules of the device:
- Flowmeters: There are three separate vertical glass tube flow indicators for O2, N2O, and AIR.
- Pressure Manometers: Three round analog pressure gauges labeled OXYGEN, NITROUS OXIDE, and AIR are located on the panel just below the work surface.
- Vaporizer Label: There is a warning on the vaporizer on the right, written in black on a yellow background: FILL ONLY WITH SEVOFLURANE.
- Other Labels: A label reading AUXILIARY OUTLETS is present to the right of the flowmeters. The O2 FLUSH button is located to the left of the pressure gauges.
Potential Failure Risk
Based on the visual analysis, the most prominent mechanical issue is the wooden supports under the drawer unit. This confirms a structural weakness in the lower part of the chassis. Due to the device being powered off, no definitive assessment can be made regarding the internal condition of the electronic systems, valves, and sensors. As it is a used and older device, potential risks include fatigue or sealing issues that may occur over time in the white corrugated hoses and black connection cables.