Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 09.05.2026
Report code: 1778330495
Minrad Sojourn Sevoflurane Anesthesia Vaporizer Analysis Report
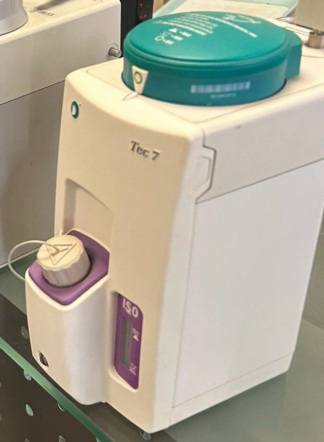
Device Identification and Field of Use
The device in the image is an anesthesia vaporizer used in anesthesia machines. This device is designed to vaporize a liquid anesthetic agent (in this case, Sevoflurane) and precisely mix it into the respiratory gas mixture to be delivered to the patient. It is used in operating room environments during general anesthesia procedures.
Brand and Model Information
According to the information clearly legible on the device body:
- Brand: Minrad
- Model: Sojourn
- Agent Type: Sevoflurane
The yellow dial on the top of the device and the “SEVOFLURANE” inscription on the body confirm that this vaporizer is calibrated exclusively for use with the Sevoflurane anesthetic agent.
Physical State and Condition
The general condition of the device appears to be used. There are minor stains and discoloration on the white plastic body in some areas. However, no serious mechanical damage such as cracks, fractures, or deep dents has been observed on the body. The yellow adjustment dial on the top and the gray concentration adjustment ring appear intact. The numbers and markings on the adjustment ring (0, 0.2, 0.4, 0.6 Vol.%, S, T) are legible. The liquid level indicator glass on the lower front of the device is intact, and the liquid level inside is clearly visible. The filling port cap (Dräger Fill) is in place, and the black tether attached to it is intact.
Mechanical and Electronic Components
The externally visible mechanical parts of the device (adjustment dial, filling cap, level indicator) maintain their integrity. The mounting bracket and locking mechanism used to connect the device to the anesthesia machine are visible on the rear side. There is no visible damage to these parts. There are no electronic displays, buttons, or cable inputs on the outer surface of the device. Such vaporizers typically operate on mechanical principles.
Labels and Markings
There are various labels and markings on the device:
- The inscription “Provided by MINRAD Sojourn SEVOFLURANE” is present on the front.
- The term “Dräger Fill” is located next to the filling cap.
- A “Vol.%” label and an arrow sign are found under the concentration adjustment ring.
- A barcode and the number “10050201” are legible on the right side of the device.
- Below the barcode, there is a circular service label marked “Bakım / Onarım” (Maintenance / Repair) with months and years indicated. Some markings are visible on this label.
- A warning symbol (exclamation mark inside a triangle) is present on the bottom of the device.
Accessories and Missing Parts
Only the vaporizer itself is present in the image. The main unit or additional hoses/adapters required for connecting the device to the anesthesia machine are not shown. The filling port cap is present. No external battery or power cable is required for the device to operate.
Conclusion and Potential Risk Assessment
The examined Minrad Sojourn Sevoflurane vaporizer maintains its physical integrity despite being used. No visible damage or deformation that would hinder operational use has been detected on the body or critical parts (adjustment dial, level indicator, filling port). The condition of the device is good. The service label on it indicates that the device has undergone maintenance in the past. Based on the visual data, no significant risk of mechanical failure has been observed. However, for the safe use of such sensitive medical devices, calibration and functional tests must be performed by an authorized technical service before use.