Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 11.10.2025
InterMed Penlon Sigma Delta Anesthesia Vaporizer Analysis Report
Overview and Product Description
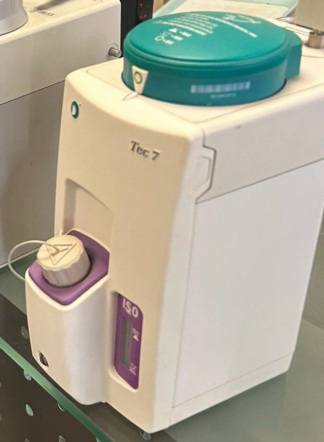
The product shown in the images is a vaporizer device used in anesthesia applications. Such devices convert volatile anesthetic liquids into gas at specific concentrations, allowing them to be administered to the patient via the respiratory tract. The specific device in the image is designed to be used only with the anesthetic substance named “Isoflurane”. The general design of the device indicates that it is manufactured for professional use in medical environments. The product is observed to have original and high-quality craftsmanship.
Brand and Model Information
Upon examining the inscriptions and logos on the product in the image, its brand and model have been clearly identified.
- Brand: InterMed Penlon
- Model: Sigma Delta
This information is clearly visible on the upper part of the device. The accuracy of the brand and model is also consistent with the product’s design details.
Physical Condition and Deformation Analysis
Despite being used, the device appears to be in very good physical condition. The following conditions were identified during a detailed examination:
- General Condition: No major scratches, dents, cracks, or broken parts are observed on the product’s casing. Its surface is clean and well-maintained.
- Color and Surface: There is no noticeable discoloration or wear on the device’s grey metallic body.
- Labels: The warning label “FILL ONLY WITH ISOFLURANE” (Sadece İsofluran ile Doldurun) on it is intact and legible. All other inscriptions and markings also retain their clarity.
Overall, it is understood that the device has been used carefully and well-preserved.
Mechanical and Technical Assessment
Based on visual analyses, the condition of the device’s mechanical components is as follows:
- Adjustment Dial: The rotary dial (VOL %) used to adjust the anesthetic substance concentration is intact. The percentage values between 0 and 5 and the pointers on it are clearly visible.
- Button and Cap: The red “PUSH ON” button located in the center of the dial is in place. The cap of the anesthetic substance filling chamber is present and attached to the device with a safety cord.
- Liquid Level Indicator: On the front of the device, the glass tube (sight glass) showing the amount of liquid inside the chamber appears intact and transparent.
There is no indication of any mechanical malfunction or missing parts in the visuals. The device is predicted to be functional in its current condition.
Accessory and Quantity Information
The images show one InterMed Penlon Sigma Delta vaporizer. No additional accessories (box, cables, user manual, etc.) other than the device’s own parts are present.
Label and Specification Information
The following information has been obtained from the labels and inscriptions present in the images:
- Substance to be Used: Isoflurane
- Concentration Range: %0 – %5
Detailed label information such as serial number, REF code, or manufacturing year, which might be found on the back or bottom of the device, is not visible in the available images.
Conclusion and Evaluation
The examined InterMed Penlon Sigma Delta anesthesia vaporizer is a medical device in very good physical condition, with no significant deformation or missing parts. The brand, model, and intended use are clearly understood from the visuals. Its mechanical components appear complete and intact. As there is no evidence of any existing malfunction in the visuals, and considering the device’s generally well-maintained state, the potential risk of malfunction is assessed as low.