Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 11.10.2025
Dräger Vapor 19.3 Anesthesia Vaporizer Analysis Report
Overview and Product Description
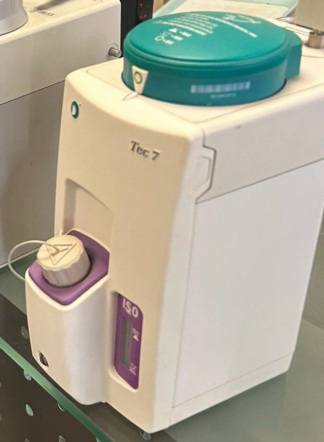
The product shown in the images is an anesthesia vaporizer used in medical anesthesia applications. The device is designed to deliver Isoflurane, a volatile anesthetic agent, to the patient at specific concentrations. The images show one vaporizer and a small accessory attached to it.
Brand and Model Information
The brand and model of the device in the images have been clearly identified. The product’s brand is Dräger, and its model is Vapor 19.3. The “Isoflurane” inscription on the device indicates that it is calibrated for use with this specific anesthetic agent. Upon examining the logo, labels, and workmanship quality, the product is assessed to be original.
Physical Condition and Deformation Assessment
The device is generally in good condition and appears used. No significant deformation such as a large scratch, dent, crack, or discoloration has been observed on its metal casing or plastic parts. The labels on it are legible and show no signs of peeling or wear. Overall, it has a well-maintained appearance.
Mechanical Component Examination
No issues were detected during the visual inspection of the device’s mechanical parts. The black control lever located on the upper part and the black knurled adjustment dial used for setting the anesthetic agent concentration appear robust. The percentage (%) values and lines on the dial are clearly legible. The connection socket and pin located at the bottom are in good physical condition.
Labels and Technical Information
Various technical details and operating instructions are present on the labels located on the body and upper part of the device. This information is as follows:
- Brand: Dräger
- Model: Vapor 19.3
- Anesthetic Agent: Isoflurane
- Factory Number (Serial No): ASNN-0263
- Manufacturer: Drägerwerk AG, Lübeck
- Place of Manufacture: Made in Germany
- Operating Instructions: The upper label of the device contains basic usage and safety warnings in English, German, and French, such as “Fill in ‘0’ position only”, “Empty for transport”, and “Do not tilt more than 45 degrees”. Additionally, a pictorial user guide is present on the device’s body.
Accessories
Other than the device itself, no accessories such as a box, extra cables, user manual, or warranty certificate are visible in the image. Only a metal chain is present at the device’s bottom connection port, preventing a small pin from falling out.
Potential Risk Assessment
Based on the examination of the images, no adverse conditions that could pose a potential risk of malfunction, such as rust, severe wear, or crushed cables, have been detected on the device. The product’s physical condition is good, and it visually shows no signs of malfunction.