Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 30.05.2026
Report code: 1780102373
IMAXEON Salient Controller Injector Device Analysis Report
Device Identification and Components
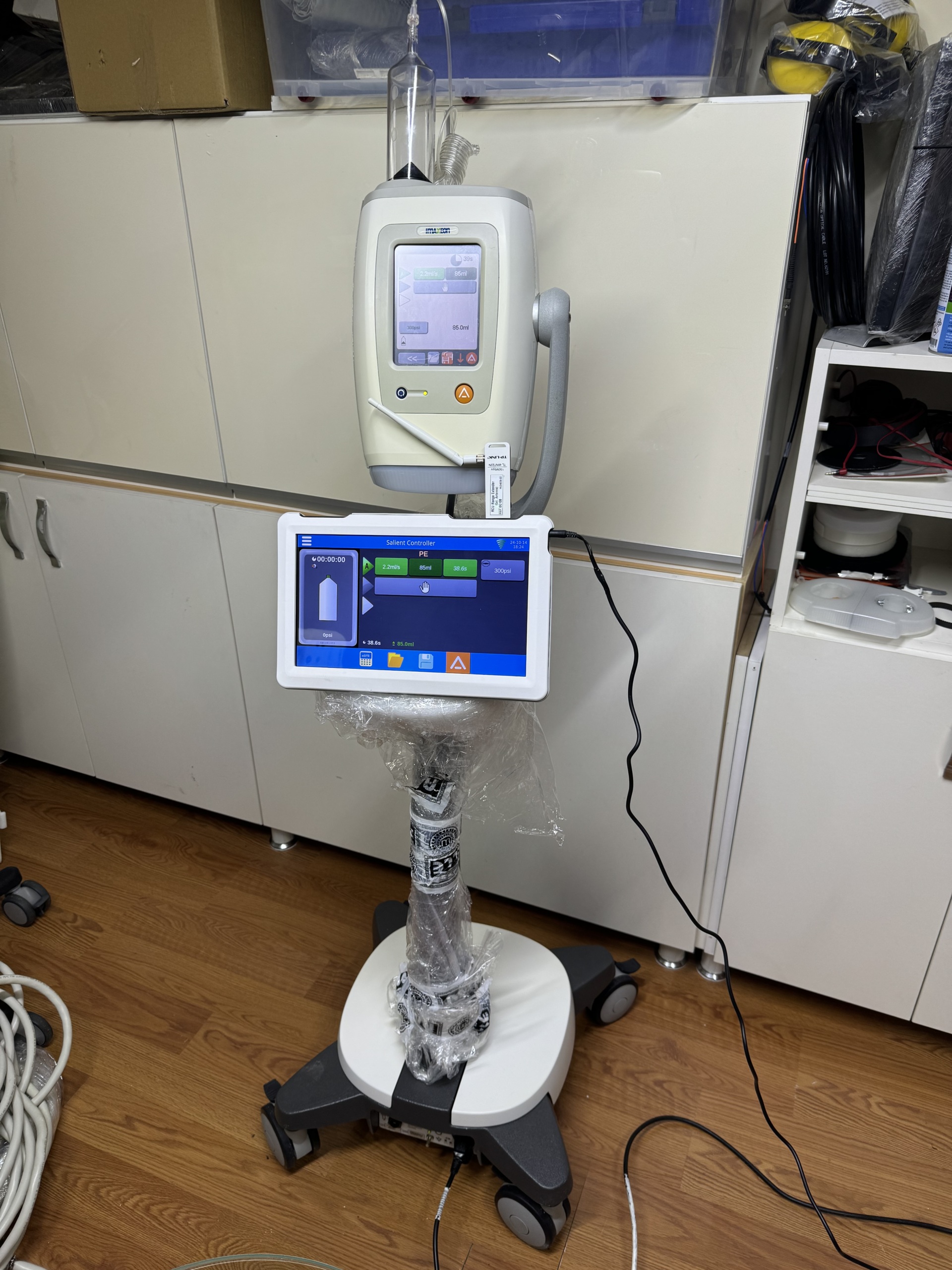
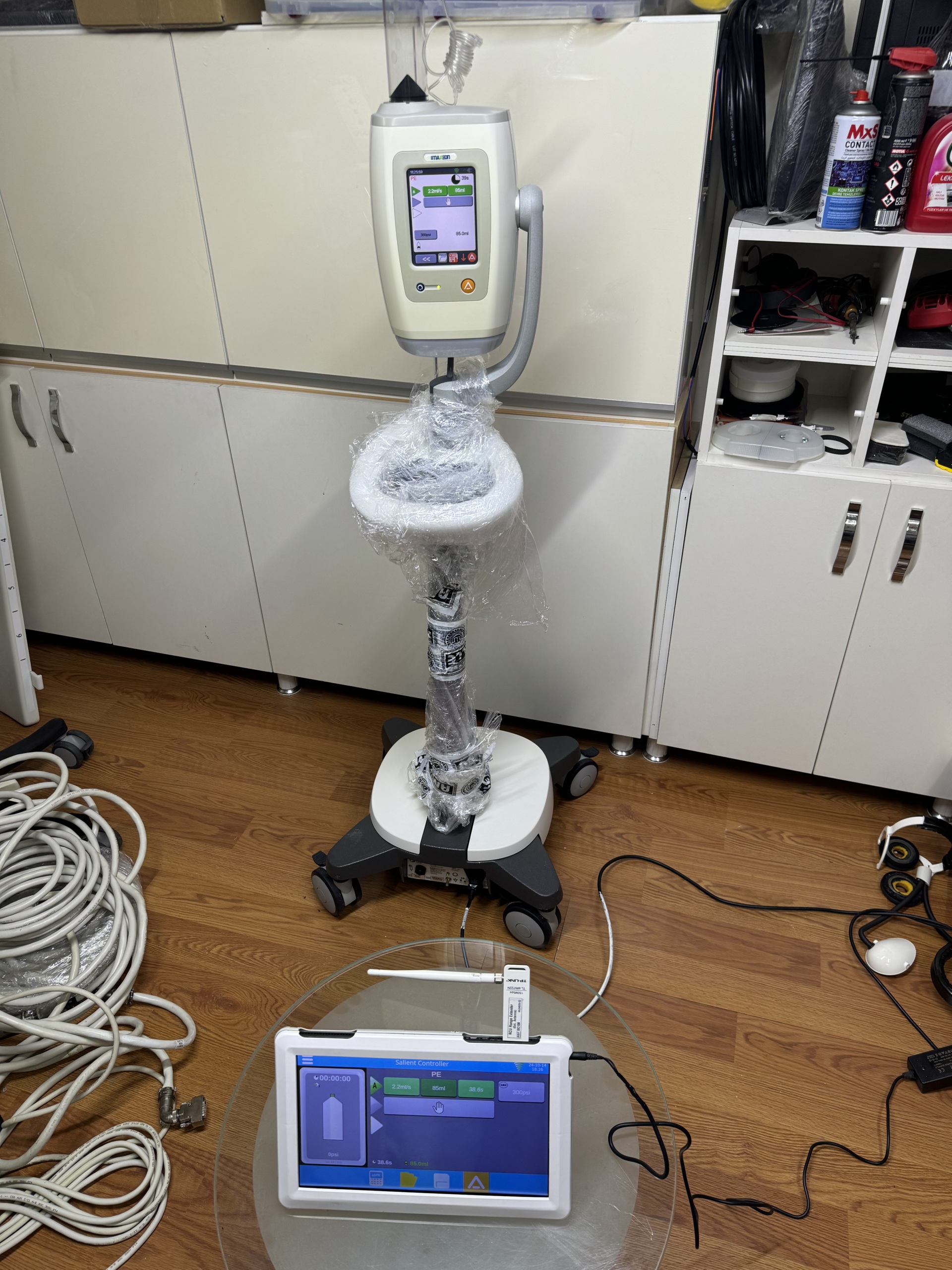
- The images show a medical injector device, a control unit belonging to this device, and a mobile stand.
- The device features a main unit designed for the controlled injection of fluids.
- The control unit is a tablet-like screen used to set and monitor the device’s operating parameters.
- The mobile stand has a wheeled structure for transporting and positioning the main unit and the control unit.
Brand and Model Information
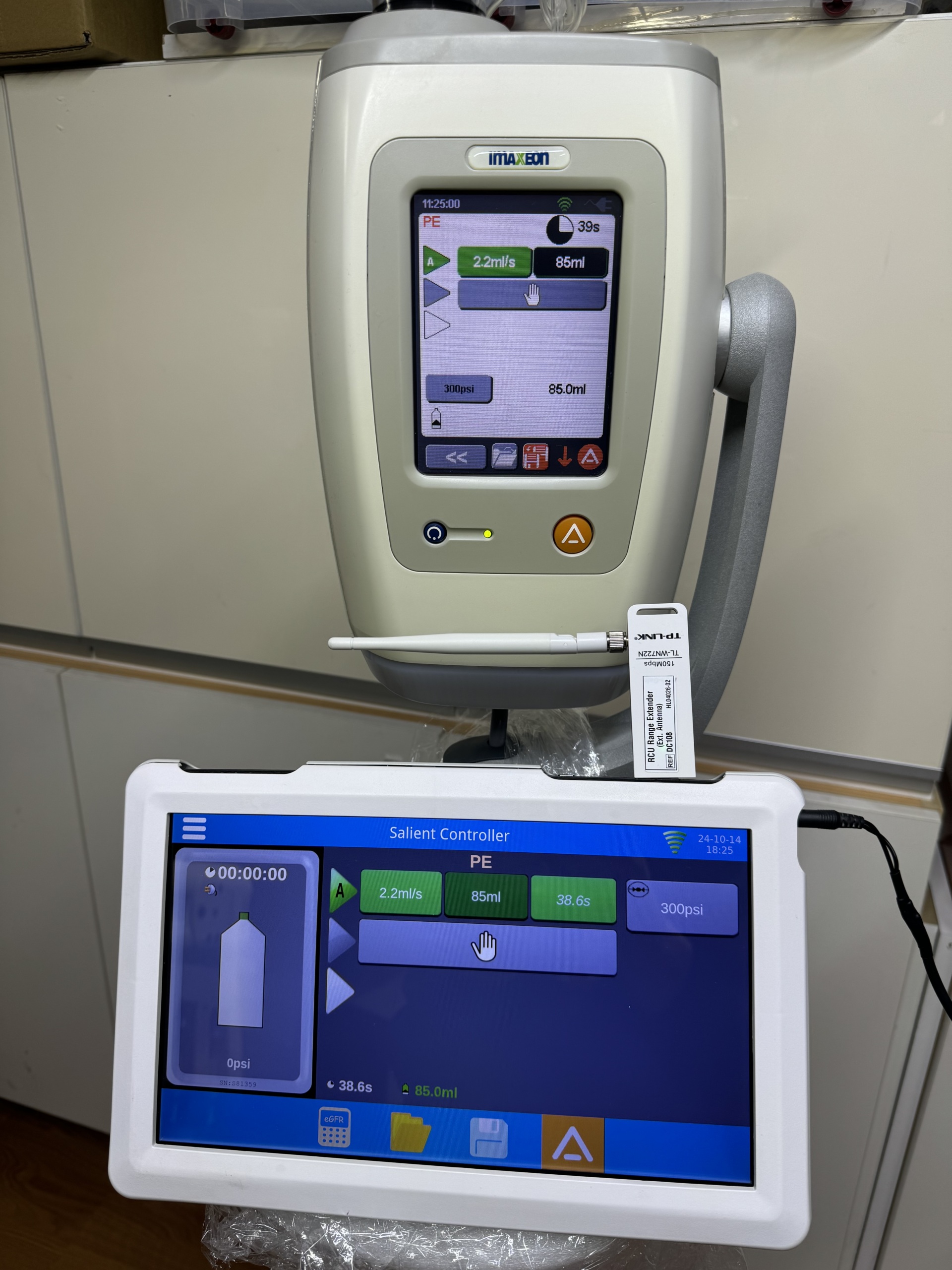
- The IMAXEON brand is clearly visible on the main device.
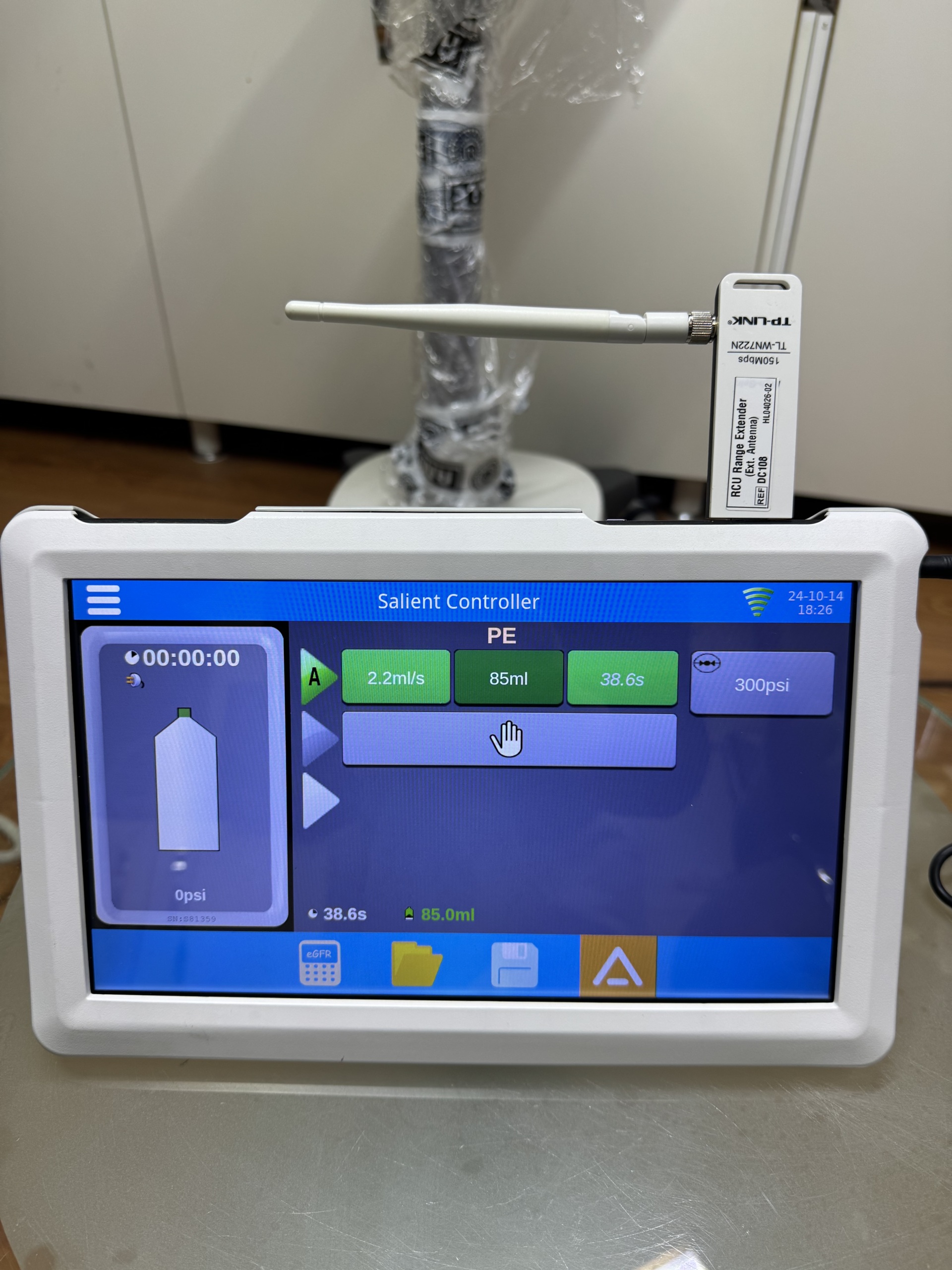
- The phrase Salient Controller appears on the control unit screen. This is the model name of the control unit.
- An external USB device attached to the unit displays the TP-LINK brand and TL-WN725N model. This device is labeled as an RCU Range Extender.
Authenticity Assessment
The quality of the logos, labels, and general workmanship on the device suggests that the product is authentic. No signs of counterfeiting have been observed.
Usage Areas
This device is an injector system designed for applications such as contrast media injection in medical imaging procedures. Parameters visible on the screen, such as “PE”, “ml/s”, “ml”, and “psi”, indicate that the device is suitable for use in specialties requiring precise fluid flow control, such as radiology, cardiology, or angiography. The TP-LINK RCU Range Extender is an accessory used to increase the wireless communication range of the device.
Quantity Information
- One (1) IMAXEON main injector device.
- One (1) Salient Controller control unit.
- One (1) mobile stand.
- One (1) TP-LINK TL-WN725N RCU Range Extender USB device.
- Various connection cables.
General Condition
The general condition of the device appears to be very good. The fact that the main pole of the mobile stand is wrapped in transparent plastic packaging suggests that the device is new, lightly used, or protected after maintenance. There is no significant wear or signs of use on the device casing or screens.
Physical Deformations
No physical deformations such as scratches, dents, cracks, discoloration, or dirt have been detected on the device’s casing, screen, or other main sections in the images. The surfaces appear clean and intact.
Mechanical Component Inspection
There are no visible mechanical issues with the mobile stand’s wheels, the main unit’s handle, or the general plastic components of the device. The protective packaging on the stand’s pole indicates that the mechanical components have been preserved.
Electronic Component Analysis
The screens on the main unit and the control unit are active and provide clear images. Information is legible on both screens. The external TP-LINK USB device is plugged into the control unit and is operational.
Accessories
- The device comes with one TP-LINK TL-WN725N RCU Range Extender USB device. This device bears the information “REF P/N: 100”, “S/N: 160M00599”, and “MAC: 00-A0-C6-00-4D-02”.
- One black cable for the device’s power connection and one black connection cable for the control unit are visible.
- There are multiple white cables gathered on the floor, but it is not clear from the images whether these belong to the device or what their exact function is.
- There are socket inputs on the device, but there is no image close enough for a detailed analysis.
- Expiration dates for the accessories are not present in the images.
Battery Status
A fully charged battery icon is visible on the control unit screen. This indicates that the control unit’s internal battery is charged or that it is connected to an external power source. No physical deformation such as swelling or leakage has been observed in the battery.
Label Information
- Serial Number: Indicated as “S/N: 160M00599” on the TP-LINK TL-WN725N USB device. This number provides the unique identity of the product and facilitates traceability.
- REF Code: Seen as “REF P/N: 100” on the TP-LINK TL-WN725N USB device. The REF code generally refers to the product’s reference part number or catalog number.
- MAC Address: Indicated as “MAC: 00-A0-C6-00-4D-02” on the TP-LINK TL-WN725N USB device. The MAC address is a unique identifier that specifies the hardware identity of network-connected devices.
Screen Analysis
The main device screen displays the time (11:25:00), “PE” mode, flow rate (2.2ml/s), injected volume (86ml), duration (39s), and pressure (300psi). There are also icons for settings and alerts. The control unit screen displays a timer (00:00:00), battery status, “A” mode, flow rate (2.2ml/s), volume (85ml), duration (38.6s), and pressure (300psi). The date and time “24-10-14 18:25” and a Wi-Fi signal strength icon are visible in the top right corner of the screen. Both screens are clear and legible, clearly showing the device’s operating parameters and status.
Dimensions and Compatibility
There is no direct information regarding the exact measurements or size of the product in the images. However, the main device and control unit resting on the mobile stand are of typical medical injector system dimensions. The wheeled stand of the device indicates that it has a structure that can be easily moved in hospital or clinical environments. The control unit is tablet-sized and works compatibly by establishing a wireless or wired connection with the main device.
Year of Manufacture
The date “24-10-14” is visible on the control unit screen. This date may indicate the period when the device’s software version or calibration was performed. Based on this information, it can be interpreted that the production period of the device is 2014 or later.
Documents
No documents such as invoices, warranty certificates, or user manuals belonging to the device are visible in the images.
Usage Duration
There is no clear information regarding the total usage hours or duration on the device’s screens or labels. The “00:00:00” timer on the control unit may indicate the duration of the current injection, but it does not specify the total usage time.
Existing Faults
No evidence of any obvious faults such as broken parts, error messages, severed cables, or missing components has been detected anywhere on the device in the images. The device screens are active and show normal operating parameters.
Potential Risk of Failure
The general condition of the device is good. No concrete situation that could pose a potential risk of failure, such as severe wear, rusting, or cable crushing, has been observed in the images. The protective packaging on the stand also suggests that the device has been well-maintained.