Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 23.02.2026
Report code: 1593095050141893
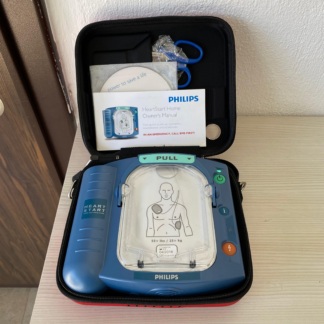
PHYSIO-CONTROL LIFEPAK 500 Automated External Defibrillator Analysis Report
Device Identification and Description
The device in the examined visuals is an Automated External Defibrillator (AED) unit designed to apply electroshock to patients experiencing sudden cardiac arrest in medical emergencies. The device is portable and contained within its special black protective carrying case.
Brand and Model
The brand and model information located on the front of the device, just above the screen, is clearly legible. The product belongs to the PHYSIO-CONTROL brand, and the model name is stated as LIFEPAK 500. Immediately below the model name, the phrase “AUTOMATED EXTERNAL DEFIBRILLATOR” defining the device’s function is present.
Physical Condition and Cosmetic Examination
The device’s outer casing is made of yellow hard industrial plastic, while the screen and keypad frame are made of black material. No prominent deep cracks or breaks were detected on the casing. However, superficial wear and dirtiness due to use are present on the carrying handle and the corners of the casing. Light scuff marks consistent with the overall condition are observed on the yellow plastic surface. The overall structure of the device maintains its integrity.
Control Panel and Screen Analysis
A small monochrome LCD screen is located in the center of the front panel. The screen is in the off position, so it is not possible to determine from the visual whether there are any pixel errors. Below the screen, three main function buttons are located:
- ON/OFF: The green button used to turn the device on and off.
- ANALYZE: The yellow button used to analyze heart rhythm.
- SHOCK: The orange/red button used to deliver a shock when necessary.
Above the screen, there are two arrow (up/down) buttons, believed to be used for menu navigation. The inscriptions on the buttons are not faded and are legible. No physical tear or collapse was detected on the membrane keypad in the visual.
Accessories and Hardware Content
The product set in the visual consists of the following components:
- 1 Physio-Control Lifepak 500 Defibrillator Device.
- 1 black, shoulder-strapped, brand-specific carrying case. The front flap of the bag features a yellow Physio-Control logo (square and telephone receiver icon). No tears are visible in the bag’s fabric, and the stitching appears robust.
- 1 quick-use instruction card is located in the transparent compartment on the inner flap of the bag.
- 1 set of electrode pads or accessory package, marked “ADULT,” is visible in sealed packaging within the internal pocket of the bag.
Label and Instruction Information
The following steps are numbered on the instruction card located in the transparent area on the inside of the bag’s top flap:
- 1. Turn on Device: Turn on the device.
- 2. Apply Electrode Pads: Apply the electrode pads (supported by a visual).
- 3. Follow Prompts: Follow the audio/visual prompts.
At the very bottom of the device’s front face, just above the carrying handle, a safety warning label is present: “WARNING: HAZARDOUS ELECTRICAL OUTPUT. FOR USE ONLY BY QUALIFIED PERSONNEL. DANGER: EXPLOSION HAZARD. DO NOT USE IN THE PRESENCE OF FLAMMABLE GASES.” These texts are legible and not faded. A small label containing a reference number (“…343-002” is legible) is visible on the shoulder strap.
Areas of Use
The PHYSIO-CONTROL LIFEPAK 500 is designed for use by pre-hospital emergency response teams, ambulances, workplaces, and in public areas for cases of sudden cardiac arrest. The “ADULT” labeled equipment in the visual package indicates that the product is configured for use with adult patients.
Potential Malfunction Risk and Deficiencies
The device’s battery is not directly visible in the visual (it is typically integrated into the casing or a module attached from the bottom), thus there is no visual evidence regarding battery presence and charge level. Although “ADULT” is legible on the pad packaging, the expiration date cannot be discerned in the visual; the medical consumables may be expired. Since the device screen is off, electronic functionality could not be confirmed. However, no mechanical damage significant enough to impede the device’s operation is visible on the outer casing.