Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 17.10.2025
Mindray BeneHeart D30 Defibrillator Analysis Report
Overview and Product Identification
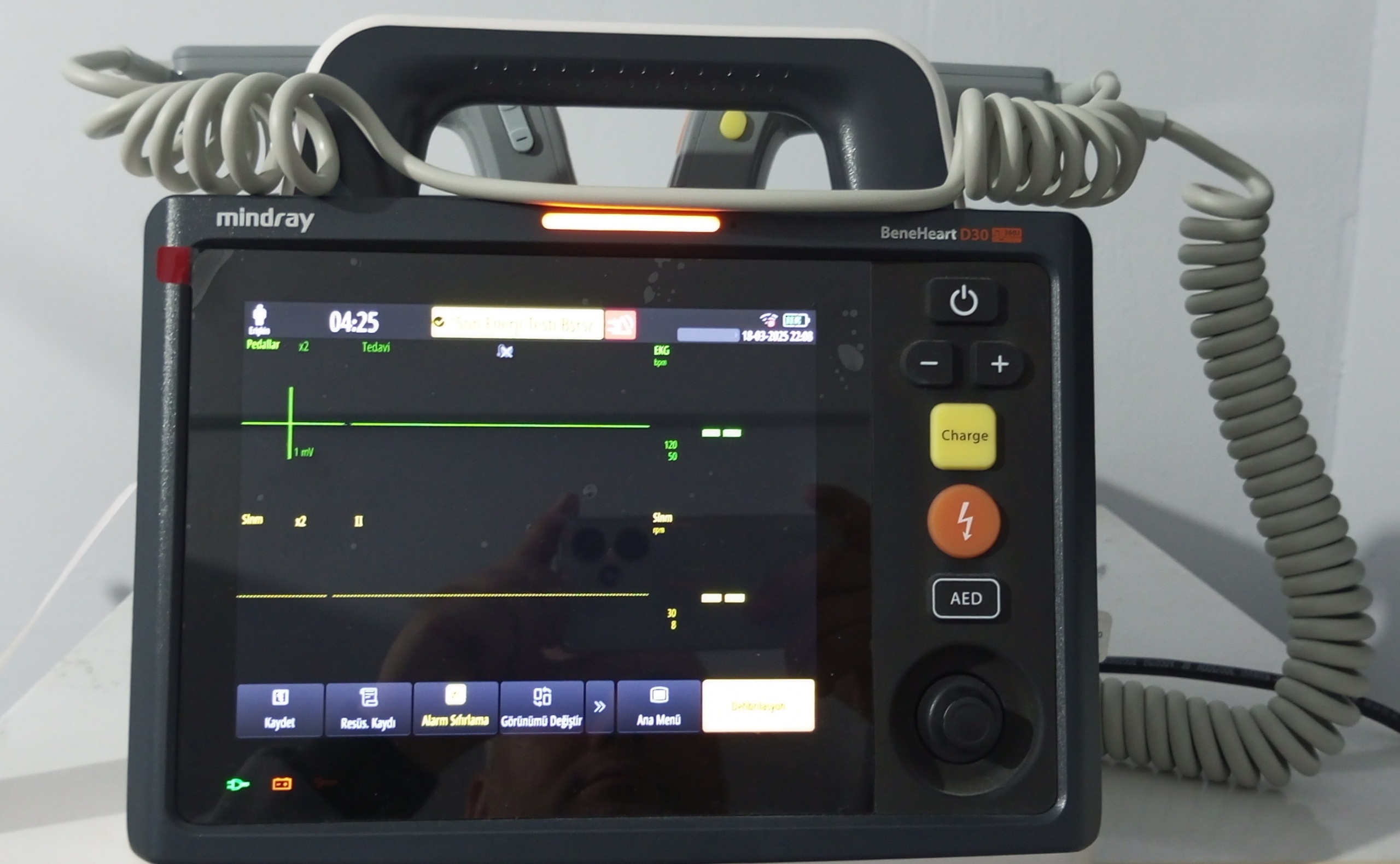
The device shown in the visuals is a defibrillator and patient monitor intended for professional medical use. The Mindray brand and BeneHeart D30 model are clearly visible on the device. The product is designed to administer electroshocks to patients in emergency situations and simultaneously monitor the patient’s vital signs (such as ECG). Its general appearance, logo quality, and the professionalism of the screen interface suggest that the device is original.
Physical Condition and Cosmetic Assessment
The device is visually in very good condition and appears to be lightly used. No significant deformation such as scratches, cracks, dents, or discoloration has been detected on the casing, screen, or accessories. The overall cleanliness of the device is notable, with no dirt or dust residues present. This indicates that the device has been used carefully and well-maintained.
Electronic and Mechanical Component Analysis
The electronic components of the device are in working condition. The visuals show that the screen is on and functional, displaying a clear and bright image. There are no pixel errors or abnormalities on the screen. The orange warning light at the top and the green status light in the printer section are active. All physical buttons (Charge, Shock, AED, control knob) and socket inputs appear intact and in place. The device’s integrated printer section also has a mechanically sound structure.
Accessory Condition
The following accessories were identified with the device in the visuals:
- External Defibrillator Paddles: A pair of external defibrillator paddles is attached to the device. Their coiled cables appear flexible and sturdy.
- ECG Cable Set: An ECG cable set with multiple electrode leads is present on the device.
- Power Cable: One power cable is connected to the device’s power input socket.
Screen Analysis and Functionality
The device’s screen provides important information about its operational status:
- The device is in “Manual” defibrillation mode and set to 200 Joules energy level.
- The date 18-03-2025 and times 04:38 and 04:25 are visible on the screen. This indicates that the device’s internal clock and battery are functioning.
- The “Alarm Off” warning indicates that the alarms have been deactivated by the user.
- Patient monitoring data such as ECG waveform and heart rate are displayed on the screen.
- The battery status indicator shows that the battery is full and the device is charging.
Label and Certification Information
No label or plate containing specific manufacturing and identification information such as the device’s serial number, lot number, or REF code was closely visible in the visuals. Therefore, this information cannot be included in the report.
Conclusion and Evaluation
Based on the examination of the visuals, the Mindray BeneHeart D30 defibrillator device was found to be in very good condition, both cosmetically and functionally. The device being powered on and operational indicates that its basic functions are active. As there are no signs of breakage, cracks, or significant wear, there is no evidence of an existing malfunction. Appearing as a complete set with its accessories, the device portrays a profile of careful use and good maintenance. With this condition, the risk of a potential malfunction is assessed as low.