Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 18.03.2026
Report code: 1773830368
Hellige SCP 913 Defibrillator Analysis Report
Device Identification and Areas of Use
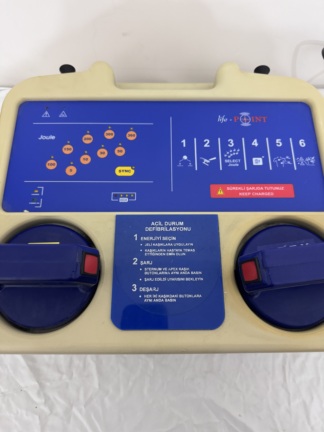
The product in the image is a defibrillator device used to apply electroshocks to restore the heart’s normal rhythm in emergencies and to monitor the patient’s ECG (electrocardiogram) data. Upon examining the logos and inscriptions on the front panel, the device’s brand was identified as Hellige, and its model as SCP 913. Such devices are typically used in hospital emergency departments, intensive care units, and ambulances. The image shows 1 main device and integrated paddles.
Overall Condition
The device is a used, older generation model. The casing shows significant yellowing and dullness due to age and use. The dirt, stains, and wear marks on it indicate that the device has been used extensively and may not have been maintained for a long time.
Physical Deformation and Mechanical Components
The following details have been observed on the device’s outer casing and mechanical parts:
- Casing: The upper parts and edges of the plastic body show black scuff marks, scratches, and dirt accumulation.
- Paint and Markings: Red paint or pen marks are present directly above the slots where the paddles are placed. On the right side surface of the device, a green tape with a black letter (likely “S”) written on it is affixed.
- Carrying Frame: A red metal carrying handle/frame surrounds the bottom and sides of the device. Paint chips and abrasions are present on the corner sections of this metal frame.
- Control Knobs: On the right side of the device, there is a large, red rotary commutator knob used to adjust the energy level. The knob maintains its physical integrity. Superficial wear due to use is observed on the membrane keypads on the left side.
- Printer Compartment: An integrated thermal printer/recorder compartment cover is located on the upper left side.
Electronic Components and Screen Analysis
Since the device is in the off position, no menus or warning messages are visible on the screens. There are two different screen areas on the front panel:
- Main Screen: The large black panel (likely CRT or an older type LCD) located on the upper left side shows no apparent cracks or fractures.
- Lower Screen: A noticeable deformation is present on the smaller, rectangular LCD screen located directly below the main screen. This area shows an irregular mark suggesting staining, delamination, or an internal crack.
Accessories and Hardware
The accessories integrated into the device in the image are as follows:
- Defibrillator Paddles: Two external shock paddles are located in their slots on the right side of the device. A red square button is on the left paddle, and a green square button is on the right paddle.
- Cables: Thick, red, spiral-shaped cables connecting the paddles to the main device are present. No external breaks or crushes have been detected in the cables.
The power cable, ECG patient cables, or other external accessories required for the device’s operation are not present in the image. Technical information such as serial number, manufacturing year, or REF code cannot be read from the labels on the device due to the distance and angle.
Potential Failure Risk
Considering the device’s overall worn condition, some potential risks exist. Particularly, the deformation observed on the lower LCD screen indicates that this screen may have lost its functionality or could cause image loss. Furthermore, due to the device’s age, there is a high risk of end-of-life for unseen internal electronic components (capacitors, internal battery, etc.). Before the device can be used in a clinical setting, it must undergo a detailed biomedical calibration and safety test.