Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 19.10.2025
Primedic Defi-Monitor Defibrillator Analysis Report
General Assessment and Product Description
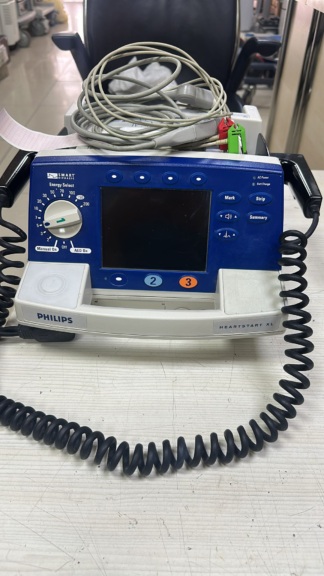
The device shown in the images is a Primedic Defi-Monitor brand and model defibrillator and patient monitor. Based on the general appearance of the device and the quality of the logos and texts on it, it is assessed to be original. This medical device is designed to administer electric shocks (defibrillation) to patients in emergency situations such as cardiac arrest, and concurrently monitor vital signs such as electrocardiography (ECG). It has a structure suitable for use by professional healthcare personnel in emergency rooms, ambulances, and hospitals.
Quantity Information and Package Contents
The breakdown of the product and accessories found in the images is listed below:
- 1 Unit Primedic Defi-Monitor main unit
- 1 Set (2 units) external defibrillation paddles, connected to the device with their cables.
- 1 Unit battery (attached to the rear section of the device)
- 1 Unit metal mounting bracket (attached to the rear of the device)
Physical Condition and Cosmetic Analysis
It is clearly visible that the device is used. Its general condition gives the impression of a device that has seen heavy use. The device’s plastic casing shows distinct yellowing and discoloration, particularly in lighter-colored areas. Surfaces generally show light scratches, dirt, and signs of wear. Abrasions are noticeable on the orange strip below the screen and in the paddle holsters. No significant breaks, cracks, or dents have been detected. The metal mounting bracket on the rear shows signs of oxidation and rust in some areas.
Mechanical and Electronic Components
Upon visual inspection, mechanical components such as the control buttons on the front panel, the carrying handle, and the holsters for the defibrillation paddles appear complete and functional. No missing or broken parts have been observed. The outer insulation of the coiled cables connecting the paddles to the device shows no visible cuts or damage. The device’s screen being on in one of the images suggests that its electronic components are at least receiving power and demonstrating basic functionality.
Accessories and Battery Condition
The primary accessory accompanying the device is the external defibrillation paddles, labeled “APEX”. These paddles are integrated with the device. A large battery pack is attached to the device in the rear section. The battery’s outer casing shows no visible swelling or leakage, but its performance and remaining lifespan cannot be commented on from the image. Accessories such as the power cable required to charge the device or other ECG probes are not present in the images.
Technical Label Information and Screen Analysis
Some information is present on the label at the back and on the front face of the device. However, due to the resolution of the images, detailed information such as the serial number cannot be clearly read. Some readable information includes:
- Battery Type: PRIMEDIC ACCU Type 06130
- Battery REF Code: ACCU 06130
- Certification: CE 0123 mark is present.
In the image where the device is turned on, an ECG waveform and various icons are visible on its screen. The text “Defistatus o.k.” on the screen indicates that the device has successfully completed its internal defibrillation test. Additionally, status indicators such as battery status and volume level are also displayed on the screen.
Potential Risk Assessment
The prominent wear and yellowing throughout the device indicate prolonged and intensive use. In such medical devices, the most critical part whose performance degrades over time is the battery. As the age and condition of the current battery are unknown, it must be tested or replaced before safe use to eliminate a potential failure risk. Furthermore, oxidation on the rear mounting bracket might suggest that the device has been in a humid environment. Although there are no clear signs of malfunction in the images that would prevent its operation, considering the device’s age and usage history, it is recommended that it be thoroughly checked and calibrated by an authorized technician before being put into clinical use.