Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 06.02.2026
Nanoom Tech HeartPlus NT-180 Automated External Defibrillator (OED) Analysis Report
Device Identification and Intended Use
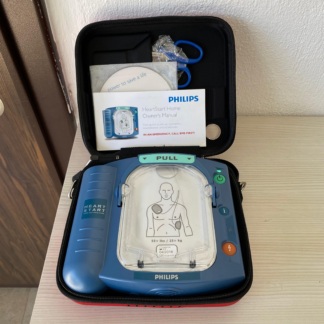
The device shown in the images is an Automated External Defibrillator (OED/AED) that delivers an electric shock to restore the heart’s normal rhythm in cases of sudden cardiac arrest. This device is designed for emergency interventions in public areas as well as for professional healthcare personnel.
Brand and Model Information
The product has been thoroughly examined in accordance with its original box, user manual, and labels on the device, as seen in the images. The identified information is as follows:
- Brand: HeartPlus (Manufacturer: Nanoom Tech Co., Ltd.)
- Model: NT-180
- Product Type: Automated External Defibrillator
Box Contents and Quantity Information
The box contents are neatly arranged and consist of the following components according to visual analysis:
- 1 HeartPlus NT-180 Defibrillator Main Unit (Red colored)
- 1 Black/Red colored original carrying case protecting the device
- 1 Sealed Silver Package (Containing Cartridge/Battery and Pads, with installation instructions on it)
- 1 User Manual
- 1 Software/Information CD (Smart Plus AED / NT-MPR)
- 1 Netmate brand sealed USB Connection Cable (Computer Cable)
- 1 White cleaning cloth or extra accessory package in transparent packaging
- 1 Black carrying strap (with metal clips)
General Condition and Cosmetic Inspection
The device and its accessories appear to be in “New” or “Open Box” condition. No scratches, cracks, impact marks, or signs of use (fading on buttons, dirt) have been detected on the main unit. Most accessories (cable, battery cartridge, cloth) are in their original sealed packaging. The device’s carrying case is clean and intact.
Mechanical and Electronic Components Analysis
- Lid and Casing: The protective lid of the device has been opened, and the interior has been viewed. There is no visible issue with the hinge mechanism. The information cards on the inner panel are clean and legible.
- Keypad and Interface: The device features an “Adult/Child” toggle switch, a shock button (with a lightning symbol), and a power button (green). The buttons appear physically intact. A status indicator LED panel is also present.
- Connection Slot: The device’s battery/cartridge slot is empty. The image shows that the battery is not installed but is in a separate package within the box. The slot is clean, and there are no signs of corrosion/rust on the pins.
Technical Label and Date Information
Critical technical data and dates have been obtained from the labels on the device box and the consumables package:
- Device Box Serial Number: NT180-S14-00007
- Date on Box: 2014.04.02 (This date indicates the device’s manufacturing date).
- Device Weight: 1.9kg (According to label information)
- Consumables (Cartridge) Package Serial Number: SN 10010-1010-1600518
- Consumables Package Date: 2018.03.03 (This date on the package could be the manufacturing or filling date).
- Battery Specification: Non-Rechargeable Lithium-ion Battery, DC 21V – 1400mAh.
Battery and Power Status
The device operates with a replaceable cartridge system, not an external charger. In the images, the “HeartPlus Cartridge” containing the battery and electrode pads is in a silver-colored, sealed package. While the unopened nature of this package supports that the product has not been used, the date factor should be evaluated under the “Potential Condition” section below.
Documents and Originality
The box contains the “User Manual” booklet, a CD, and all labeling that gives the impression of warranty/originality. Nanoom Tech’s logos and part compatibilities indicate that the product is 100% original.
Current Status and Potential Risk Assessment
Although the product is in excellent physical condition, there is an important aspect that buyers should consider due to the nature of the device:
Expiry / Shelf Life Warning: The box label in the images indicates 2014, while the sealed battery/cartridge package indicates 2018. Defibrillator batteries and adhesive electrode pads have a specific shelf life. Considering the time elapsed since the manufacturing date, it is technically highly probable that the battery inside the sealed package has depleted its energy or that the electrodes have lost their properties (dried out). Although the device body is intact, a new, up-to-date battery/cartridge set may need to be purchased to make it ready for use.