Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 15.09.2025
Nihon Kohden TEC-5521K Defibrillator Analysis Report
Overview and Device Identification
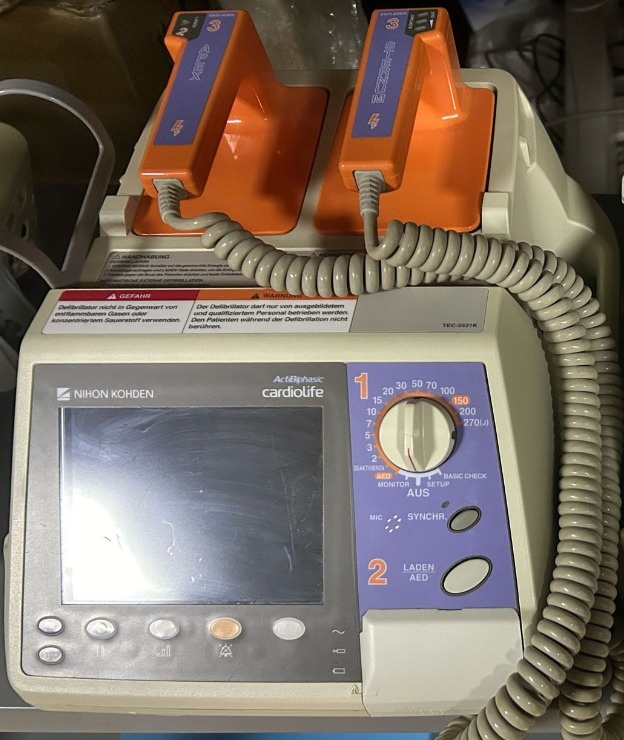
The product shown in the images consists of one Nihon Kohden TEC-5521K model defibrillator and two external defibrillation paddles integrated into the device. The device is a medical instrument used to restore the heart to its normal rhythm by delivering an electrical shock in emergencies. Its overall appearance suggests that it is a used product. The logo and writings observed on the product indicate that it is an original device.
Brand and Model Information
The brand and model of the device have been clearly identified in the images. The NIHON KOHDEN brand and cardiolife series are clearly visible on the device’s front panel. Additionally, the TEC-5521K model code is present on the label on the control panel.
- Brand: Nihon Kohden
- Series: cardiolife
- Model: TEC-5521K
- Technology: ActiBiphasic
Physical Condition and Deformation
The device is generally in a used condition and shows visible signs of wear. Yellowing and discoloration, particularly on the white plastic parts of the device’s casing, are noticeable and have occurred over time. The screen surface has multiple scratches and stains. Slight dirt accumulation has been observed on the device’s buttons and control panel. However, no breaks, cracks, or serious impact marks were found on the casing.
Mechanical and Electronic Components
Upon visual inspection of the device’s mechanical parts, the function selection knob and other control buttons appear to be in place and physically sound. The defibrillation paddles fit properly into their slots, and the connection cables maintain their coiled structure. No crushing, cuts, or exposed wires are visible on the cables. The device’s screen is off, therefore, it is not possible to determine if the electronic components are functioning or if there are any error codes on the screen.
Accessories and Label Information
In the images, no other accessories (power cable, ECG electrodes, etc.) are visible apart from the two defibrillation paddles attached to the device.
A label containing German warnings is located on the front surface of the device. This label includes the following information:
- GEFAHR (Danger): It is stated that the defibrillator should not be used in environments containing flammable gases or concentrated oxygen.
- WARNUNG (Warning): It is stated that the defibrillator should only be used by trained and qualified personnel, and the patient should not be touched during defibrillation.
No information regarding a serial number or year of manufacture could be identified on the labels in the images.
Potential Risk Assessment
The yellowing on the device’s outer casing indicates long-term use or the age of the product. Scratches on the screen may slightly hinder the readability of critical information that should be displayed when the device is operational. Although no obvious malfunction was detected based on visual evidence, due to it being a used medical device, it is crucial that internal components such as calibration and battery are checked by a professional technical service. Considering the current condition of the cables and mechanical components, no serious potential malfunction risk has been observed.