Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 11.03.2026
Report code: 1773242918
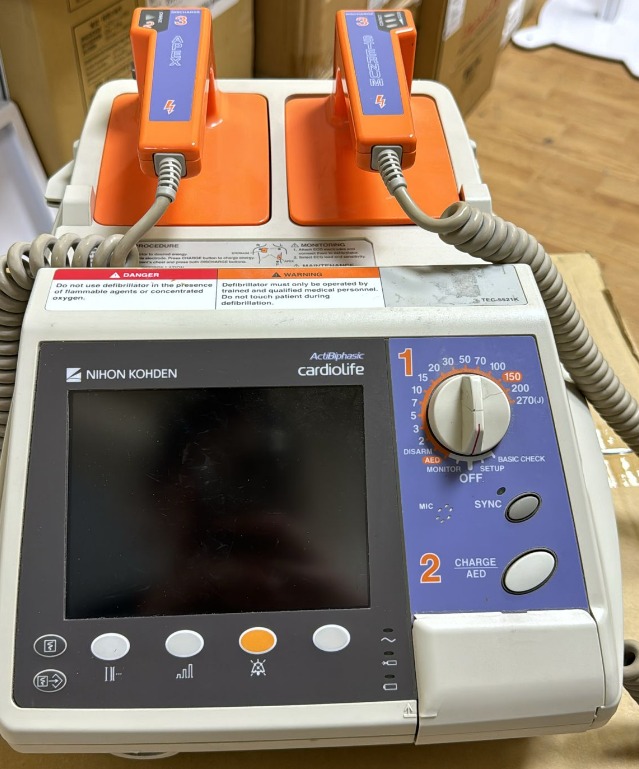
Nihon Kohden Cardiolife TEC-5521K Defibrillator Analysis Report
Device Identification and Basic Information
The product shown in the images is a defibrillator device used to deliver an electric shock to the heart in emergencies. The brand and model were identified by examining the device’s front panel and its labels.
- Brand Nihon Kohden
- Series ActiBiphasic cardiolife
- Model TEC-5521K
Areas of Use
This device is used in hospital emergency departments, intensive care units, operating theaters, and ambulances to intervene in cardiac arrest situations. Examination of its control panel reveals that it has manual energy selection and AED (Automated External Defibrillator) modes. These features indicate that the device is designed for both professional manual use and semi-automatic use.
Physical Condition and State
The device is in used condition, and its outer casing shows noticeable signs of use.
- Casing General yellowing and discoloration are observed on the white plastic body.
- Deformation On the upper part of the device, immediately next to the warning label on the right side, there is a noticeable trace of a removed label, scratching, and adhesive residue. Minor superficial scratches are present on the casing.
- Screen The screen on the front of the device is in the off position. No cracks or breaks are visible on the screen glass, but slight stains are present on its surface.
- Mechanical Components The energy selection dial, buttons, and paddle receptacles maintain their physical integrity. No broken or missing external mechanical parts have been detected.
Paddles and Cables
Two external defibrillator paddles are located in the upper slot of the device.
- Apex Paddle It has an orange plastic casing. It features a charge (Charge) button and a shock (Discharge 3) button. It is physically intact.
- Sternum Paddle It has an orange plastic casing. It features a contact quality indicator (Contact) LED area and a shock (Discharge 3) button. It is physically intact.
- Cables The gray spiral cables belonging to the paddles are connected to the device. No breaks, crushes, or external insulation tears have been observed on the cables. Slight dirt is present on the cable surfaces.
Control Panel and Interface
The controls on the device’s front panel are as follows
- Energy Selection Dial Energy levels between 2 and 270 Joules can be selected. Additionally, the dial includes OFF, SETUP, BASIC CHECK, MONITOR, AED, and DISARM modes.
- Buttons Below the dial, there is a Charge/AED button and a Sync button. Immediately below the screen, there are four unnamed function (soft key) buttons used for menu navigation.
- Indicators In the lower right corner of the screen, there are icons and LED areas indicating mains power, battery status, and system warnings.
Labels and Warnings
On the upper part of the device, immediately below the paddles, there are various warning and usage instruction labels in English.
- Danger It is stated that it should not be used in environments containing flammable agents or concentrated oxygen.
- Warning It states that the device should only be used by trained and qualified medical personnel and that the patient should not be touched during defibrillation.
- Operation Procedure There is a short text explaining the energy selection, charging, and shocking steps, along with a visual diagram showing the placement of electrodes on the body.
Quantity and Authenticity Assessment
In the images, 1 main device and 2 paddles integrated into the device have been identified. Upon examining the device’s casing structure, the print quality of brand logos, the placement of labels, the fonts used, and the texture of the plastic material, the product is assessed to be an original Nihon Kohden production.
Potential Malfunction Risk
During visual inspection, no major damage such as a broken part, a severed cable, or a physical impact mark that would prevent the device from operating has been detected. The yellowing on the outer casing and the traces of removed labels are purely cosmetic flaws. The fact that the spiral cables, which are critically important for defibrillators, appear intact and integrated, is a positive aspect. The overall physical condition of the device is good for a used medical device.