Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 13.08.2025
M3 Tel Med C Pen ECG Monitor M3 Analysis Report
Device Identification and Applications
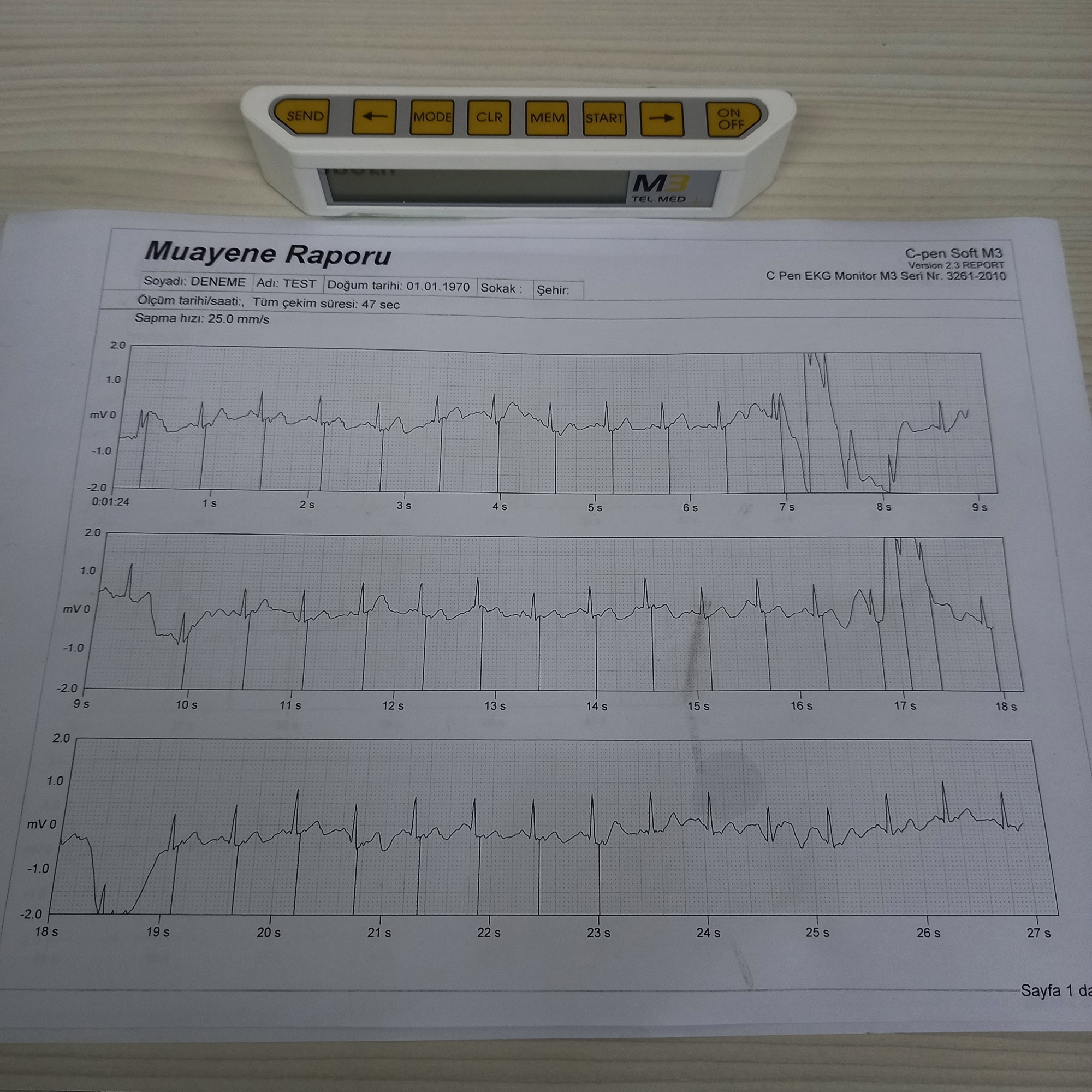
The images show a portable ECG (Electrocardiography) monitor device and its accessories. This product is used to measure, record, and report ECG data. The portable nature of the device indicates that it is suitable for on-the-spot measurements and patient monitoring. With the accompanying software, it is possible to examine and report the measurement results in detail on a computer.
Brand and Model
Following examinations of the device and software in the images, the brand and model information have been clearly identified.
- Device Brand and Model: M3 Tel Med C Pen ECG Monitor M3
This information has been confirmed by both the “M3 TEL MED” logo on the device and the phrase “C Pen EKG Monitör M3” on the “Examination Report” document. Additionally, the text “TEL.MED C-pen-soft M3” is present on the software CD.
General Condition and Authenticity
The device, its accessories, and the carrying case are in very good condition overall. From its appearance, it is understood to be either unused or very lightly used. The quality, print clarity, and consistency of the logos, text, and labels on the product, its accessories, and documents suggest that the product is authentic. No signs of wear or poor quality have been observed.
Item Count
A list of the product and accessories shown in the images is provided below:
- 1 x M3 Tel Med C Pen ECG Monitor
- 1 x black protective sleeve for the device
- 1 x black plastic carrying case
- 1 x “C-pen-soft M3 Version 2.3 REPORT” software CD
- 1 x “LogiLink” brand USB adapter (dongle)
- 1 x USB cable
- 1 x pack of ECG electrode pads
- 1 x printed “Examination Report” document
Physical and Mechanical Assessment
No scratches, cracks, dents, or discoloration are observed on the device’s white plastic casing and screen. The product is clean and free of deformation. The yellow buttons on the device (SEND, MODE, CLR, MEM, START, ON/OFF) and the directional keys are physically intact and undamaged. The latches and hinge of the black plastic carrying case appear sturdy and have no significant deformation.
Accessory and Box Content Analysis
All of the product’s accessories are present inside the carrying case. The device, protective sleeve, software CD, connection accessories, and electrodes are presented as a complete set. No parts are thought to be missing.
- Software CD: The CD labeled “TEL.MED C-pen-soft M3 Version 2.3 REPORT” contains the device’s reporting software. No significant scratches are visible on the CD’s surface.
- Connection Accessories: There is one USB cable and a “LogiLink” brand USB adapter. The cables have no damage such as crushing or peeling.
- Electrodes: There is a quantity of ECG electrode pads inside the package. The text “AC-REGELECT” is legible on the package label. The fact that the package is sealed indicates that the electrodes are unused. However, it should be noted that such medical consumables may have an expiration date; the expiration date information cannot be determined from the image.
Label and Document Information
The technical information on the device, documents, and accessories shown in the images is detailed below.
- Serial Number: The “Examination Report” document contains a code “Seri Nr: 3261-2010”.
- Software Version: The information “Version 2.3 REPORT” is present on the CD.
The report document contains fields filled for testing purposes, such as “Soyadı: DENEME Adı: TEST” and “Doğum tarihi: 01.01.1970”. This indicates that the document is a sample printout. Official documents such as an invoice or warranty certificate were not found in the images.
Potential Malfunction Risk
In the visual inspection of the device and its accessories, no signs of breakage, burns, crushing, or significant wear were found. Therefore, there is no visible risk of malfunction. The overall condition of the device is quite good. Only, it should be considered that the ECG electrode pads may have lost their effectiveness, as they are disposable consumables subject to an expiration date. This situation does not pose a risk related to the device itself.