Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 17.10.2025
Zoncare ECG Device Analysis Report
General Assessment
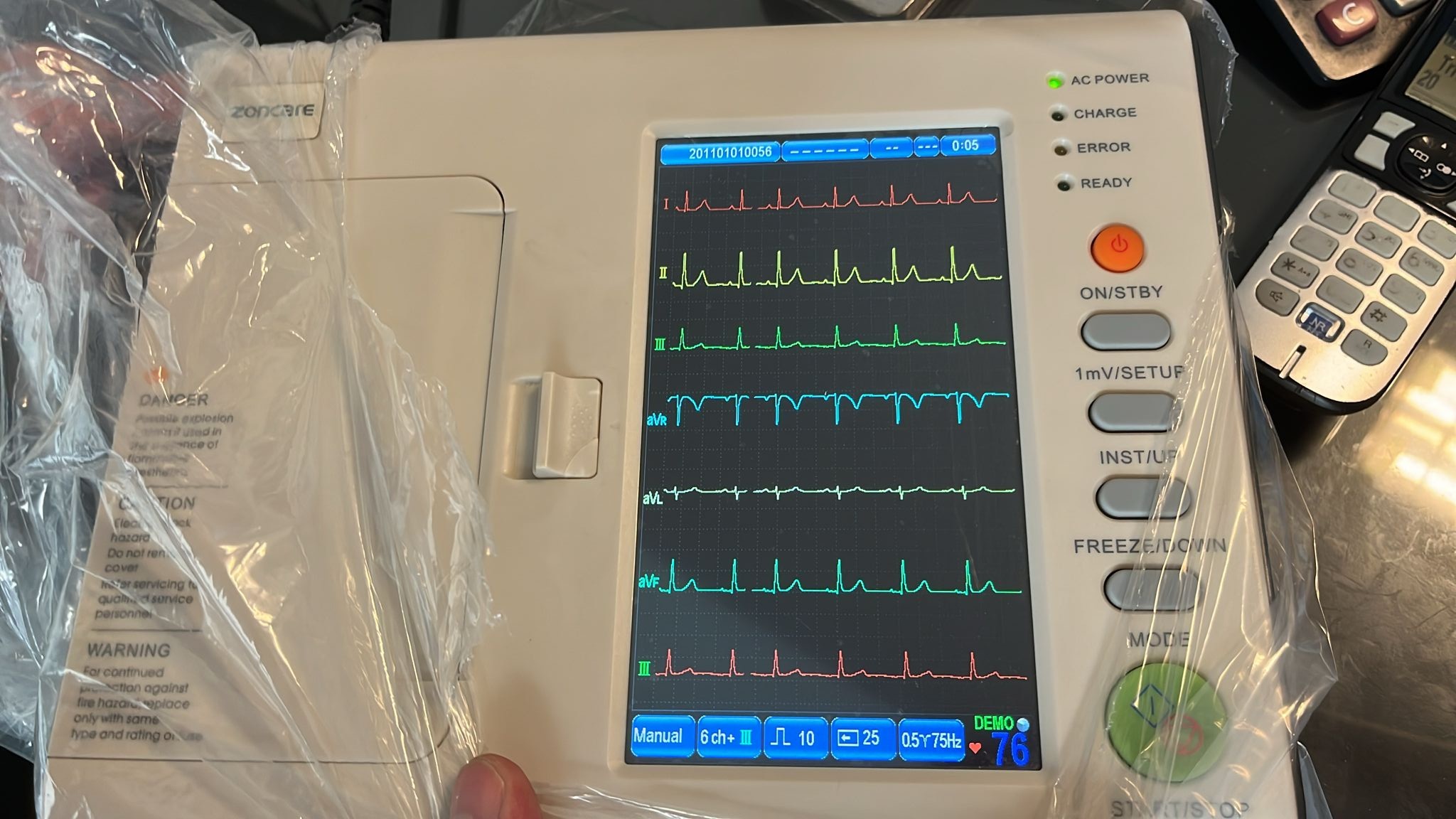
The device shown in the images is an electrocardiography (ECG) device. The brand of the product is Zoncare, as clearly visible on the front panel of the device. The device is designed for use in medical environments to monitor and record the heart’s electrical activity. The presence of the packaging bag still on the device in the image suggests that it is a new or very lightly used product. All information stated in the report has been obtained solely through the analysis of the provided images.
- Device Type: Electrocardiography (ECG) Device
- Brand: Zoncare
- Quantity: 1 main device
Device Condition and Physical Analysis
The overall condition of the device is very good. The presence of its protective plastic packaging indicates that the product might be new or a display item. No physical deformation has been detected.
- Condition: As the product still has its protective film, it is evaluated as “new” or “like new”.
- Physical Deformation: No defects such as scratches, dents, cracks, discoloration, or dirt have been observed on the device’s casing, screen, or buttons.
- Mechanical Components: All buttons on the device, the printer paper compartment cover, and other plastic components appear to be completely intact and functioning smoothly.
- Accessories: No cables, electrodes, or other accessories belonging to the device are visible in the images. Only the main device has been analyzed.
Electronic Components and Screen Analysis
The electronic components of the device are in working order. The screen is clear and bright, and the LED indicators on the device are also active. This indicates that the basic electronic functions of the device are operational.
- Power Status: In the first image, “AC POWER” and “READY” LED lights are seen lit in green. This indicates that the device is connected to power and ready for use.
- Screen: The device’s color screen is functioning without issues. There are no pixel errors or any abnormalities on the screen.
- Software Interface: The screen displays a “DEMO” mode showing ECG waveforms and a “Function Setup” menu for device settings. It is understood that the menu language can be selected as Chinese and English.
- Information on Screen:
- While operating in “DEMO” mode, the device displays a heart rate of 76.
- Technical settings such as Filter (0.5~75Hz), acquisition mode (Manual), and number of channels (6 ch + 1R) are visible on the screen.
- The date setting in the device’s software shows the year “2011”. This may not represent the manufacturing date but could be the software’s default setting.
Potential Risk Assessment
Based on the examinations in the images, no signs of malfunction or serious wear that could pose a potential risk of failure have been observed on the device. The fact that the product has been kept with its protective packaging indicates that its physical condition is very good, and it is anticipated to operate without issues in its current state.