Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 31.10.2025
Mortara ELI 150c ECG Device Analysis Report
Overview and Device Description
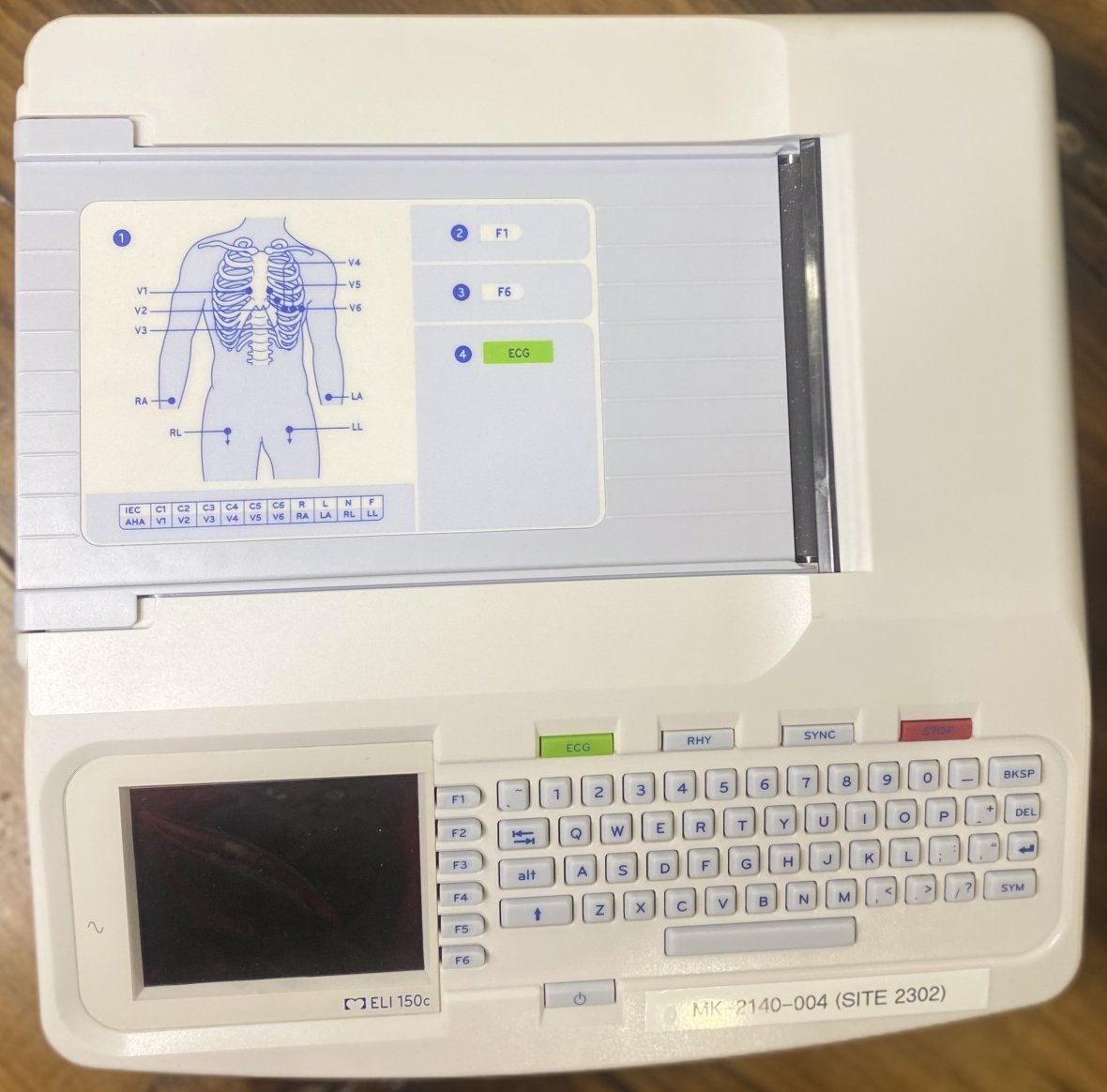
The visuals show an electrocardiography (ECG) device intended for medical use, along with various accessories belonging to this device. The product has a compact structure designed to perform ECG measurements and print the results. The device features a keyboard, a screen, and a diagram showing ECG electrode placement. The product is seen with its original box and unopened accessory packages.
Brand and Model Information
The model information ELI 150c is clearly legible on the label located below the device’s screen. The Mortara brand logo is visible on the packaging of the patient cable among the accessories. In light of this information, the product has been identified as a Mortara ELI 150c model ECG device.
Usage Areas and Functionality
This device is used in healthcare institutions to record and analyze the heart’s electrical activity and to generate ECG reports. Its portable design makes it suitable for use in various environments. The accessories, including ECG paper, a patient cable, and electrode pads, constitute the necessary components for the device to perform a fully functional ECG recording.
Quantity Information
The breakdown of the product and accessories shown in the visuals is listed below:
- 1 unit Mortara ELI 150c ECG Device
- 1 patient cable
- 1 USB cable/transfer adapter
- 1 power cable package (containing at least two cables)
- 1 box of thermal ECG printer paper
- 1 package of electrode skin prep pads
- 1 envelope labeled “Contains Important Documents”
- 1 original carrying box
General Condition and State
The device’s general condition is very good, appearing as an almost unused or refurbished product. No discoloration or yellowing has been observed on its white casing. Both the device itself and its accessories are clean and well-maintained. A large portion of the accessories are in their original, sealed packaging. This suggests that the product has been used very little or not at all.
Physical Deformation
Upon detailed examination of the device’s casing and surfaces, no scratches, dents, cracks, or impact marks were found. The surfaces are clean and smooth. The device’s screen is off and has a dark surface that might be a protective layer. No damage is observed on this surface either.
Mechanical and Electronic Components
The keys on the device’s keyboard are clean, their inscriptions are legible, and they show no signs of wear. The printer paper tray cover and other mechanical parts appear to be in place and intact. The power input, USB ports, LAN port, and other sockets on the rear of the device are clean and show no signs of physical damage.
Detailed Analysis of Accessories
- Power Cables: Two power cables, one blue and one black, are found in a “StatTech.com” branded bag.
- Patient Cable: A gray, multi-lead ECG patient cable is present in a transparent “AMMEX Latex Free Patient Cable” and “Mortara” logo-branded packaging.
- Thermal Printer Paper: A box branded “Cardiocore Thermal ECG Printer Paper” contains 10 packs of printer paper. The box bears the code REF A10091-30.
- Other Accessories: One package of “Electrode Skin Prep Pads” and a black USB adapter are found in a “Cardiocore” logo-branded bag.
Label Information
- Label on the front of the device: MK-2140-004 (SITE 2302)
- Technical information on the rear of the device: 100-240V ~ 50/60Hz 110VA
- Reference code on the ECG Paper box: REF A10091-30
No clear information regarding a serial number or lot number could be identified in the visuals.
Potential Malfunction Risk
Based on the evidence in the visuals, no current or potential malfunction risk has been observed in the device. The device’s physical condition, the fact that its accessories are packaged and new, and the absence of any signs of wear or damage indicate a high potential for the product to offer long-lasting and trouble-free use.