Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 29.11.2025
ArmoLine AL-C411 Single Channel Interpretive ECG Device Analysis Report
Device Identification and Information
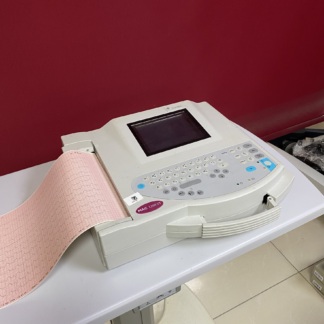
As a result of a detailed examination of the visuals, the device has been identified as an Electrocardiography (ECG) device, which is a medical measurement instrument. The “ArmoLine” brand is clearly legible on the front of the product. An analysis of the device’s rear label shows that the full name of the product is “SINGLE CHANNEL INTERPRETIVE ECG DEVICE”.
In light of the label information, the product’s brand has been determined as ArmoLine, and its model as AL-C411. Furthermore, the label states that the importing company is “Medimport Sağlık Ürünleri Ltd. Şti.” and the exporter/technology provider is “Vcomin Technology Limited”. The place of manufacture is indicated as China (P.R.C.).
Areas of Use
This device is used in healthcare institutions, emergency services, polyclinics, and family health centers to record the electrical activity of patients’ hearts. The phrase “Single Channel Interpretive” indicates that the device can perform basic automatic interpretation when generating ECG output and records the process onto paper via a single channel. Its portable design also makes it suitable for use in mobile healthcare services.
General Condition, Authenticity, and Cosmetic Examination
The cosmetic condition of the device is excellent. The blue protective gelatin on the LCD screen of the front panel, which has not yet been removed, strongly indicates that the device is an unused (new) or very lightly used product, freshly taken out of its box. No yellowing, discoloration, dirt, or stains have been observed on the plastic casing. The quality of workmanship, logo printing, and the neatness of the labels confirm that the product is an original ArmoLine device.
Physical Deformation and Mechanical Components
There are no cracks, dents, or deep scratches on the device casing, its corners, or its base. The cover of the thermal printer compartment and the paper output point located on the upper left side of the device appear intact. The ventilation grilles on the rear surface are clean, and no dust accumulation has been observed. The black rubber feet that provide grip for the device on the surface are in place and show no signs of wear.
Keypad and Screen Analysis
The device’s front panel features a membrane-type keypad. There is no fading or wear on the keys. The keypad includes the following function groups:
- Power Management: On/Off button and mains/battery status LEDs.
- Navigation and Settings: Menu, File, Freeze.
- Function Keys: F1 to F5 (for functions such as Mode, Reset, Lead, etc.).
- Operational Keys: Patient data entry, directional keys, OK, and Print (Run/Stop) button.
As the device is in the off position, no comment can be made regarding the operational status of the screen; however, physically, there are no breaks or impact marks on the screen surface.
Label Information and Technical Details
The data obtained from the main label and battery label on the rear surface of the device are as follows:
- Product Name: SINGLE CHANNEL INTERPRETIVE ECG DEVICE
- Brand/Model: ArmoLine / AL-C411
- Serial No: A “Serial No” field is present on the label (partially covered/unreadable with red in the visual).
- Certification: The CE 0123 mark is present, indicating the device’s conformity to medical device standards.
- Warnings: A warning regarding explosion risk in the presence of flammable anesthetics and fire hazard (fuse type warning) is present in English on the front panel (DANGER/WARNING).
Battery and Power Status
Battery information is clearly stated on a separate cover at the back of the device. The device has an internal rechargeable battery. The technical specifications of the battery are as follows:
- Battery Type: Ni-MH BATTERY (Nickel-Metal Hydride)
- Features: RECHARGEABLE
- Voltage/Capacity: 12V 1500mAh
Accessories and Connections
In addition to the main unit, a connection socket with a grey tip and yellow cable, observed to be attached to the left side of the device, is present in the visual. This component is assessed to be an ECG lead cable (patient connection cable). Furthermore, the power cable or adapter is not directly and fully visible in the visual, but the presence of the patient cable’s socket indicates that the accessory is present.
Quantity Identified: 1 ECG Main Device and 1 cable socket attached to the device.
Fault and Risk Assessment
As a result of visual analysis, no breaks, missing parts, or physical damage were found on the device. Due to its “new” condition, the risk of mechanical failure is considered low. However, the device’s battery uses Ni-MH technology. If the device has been stored on a shelf for a long time without charge, the possibility of the battery losing performance (due to its chemical structure) should be noted as a potential risk. Otherwise, the device appears ready for use and trouble-free.