Ready For Sale
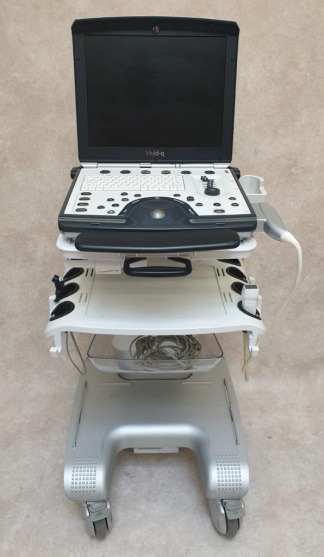
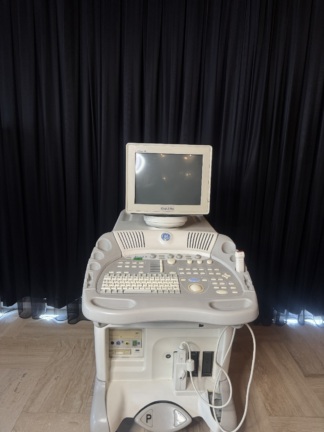
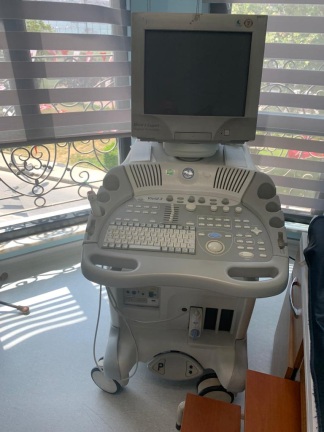
Secondhand Edan Acclarix LX3 Echocardiography Machine
Price: USD$ 20.000,00 Approx: 900.000,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 03.04.2026
Report code: 1775220461
EDAN Acclarix LX3 Ultrasonography Device Analysis Report
Device Identification and Areas of Use
The product shown in the images is an ultrasonography (ultrasound) system used for medical diagnosis. Considering the cardiac (Adult Card) and fetal (Fetal Ech) indications present in the screen images, along with the probe types found next to the device, it has been determined that the device is suitable for use in cardiology, gynecology and obstetrics, and general imaging branches.
Brand and Model Information
Based on the indications found on the main monitor casing of the device, under the touch control panel, on the rear label, and in the user manual, the brand has been definitively identified as EDAN and the model as Acclarix LX3.
Quantity and Accessory Information
The hardware and accessories identified in the images are listed below.
- 1 EDAN Acclarix LX3 main ultrasonography unit
- 1 P5-1Q model ultrasound probe
- 1 C5-2Q model ultrasound probe
- 1 P7-3Q model ultrasound probe
- 1 English user manual (Advanced Volume, Version 1.3)
General Condition and Physical State
No scratches, cracks, breaks, or discoloration have been observed on the device’s outer casing, main monitor, or touch control panel. Plastic components and screen surfaces are clean. No signs of physical deformation, crushing, or wear have been found on the cables or bodies of the ultrasound probes. The device generally appears new and well-maintained.
Originality Assessment
The print quality of the brand logos on the device, the detailed technical data on the manufacturer’s label on the rear panel, the UDI barcode system, the CE certification mark, and the presence of the device’s original user manual indicate that the product is an original medical device.
Label and Technical Information
The data on the manufacturer’s label located on the rear of the device is presented below.
- Product Name Diagnostic Ultrasound System
- Model Acclarix LX3
- Manufacturing Date 2023-08-25
- Serial Number (SN) 560522-M23816170001
- Maximum Weight 55 kg
- Power Requirement 100V-240V~ 50Hz/60Hz 2.5A-1.2A
- Protection Against Electric Shock Class I
- Certification CE 0123
- UDI Information (01)06944413813298, (11)230825, (21)560522-M23816170001
Screen and Electronic Components Analysis
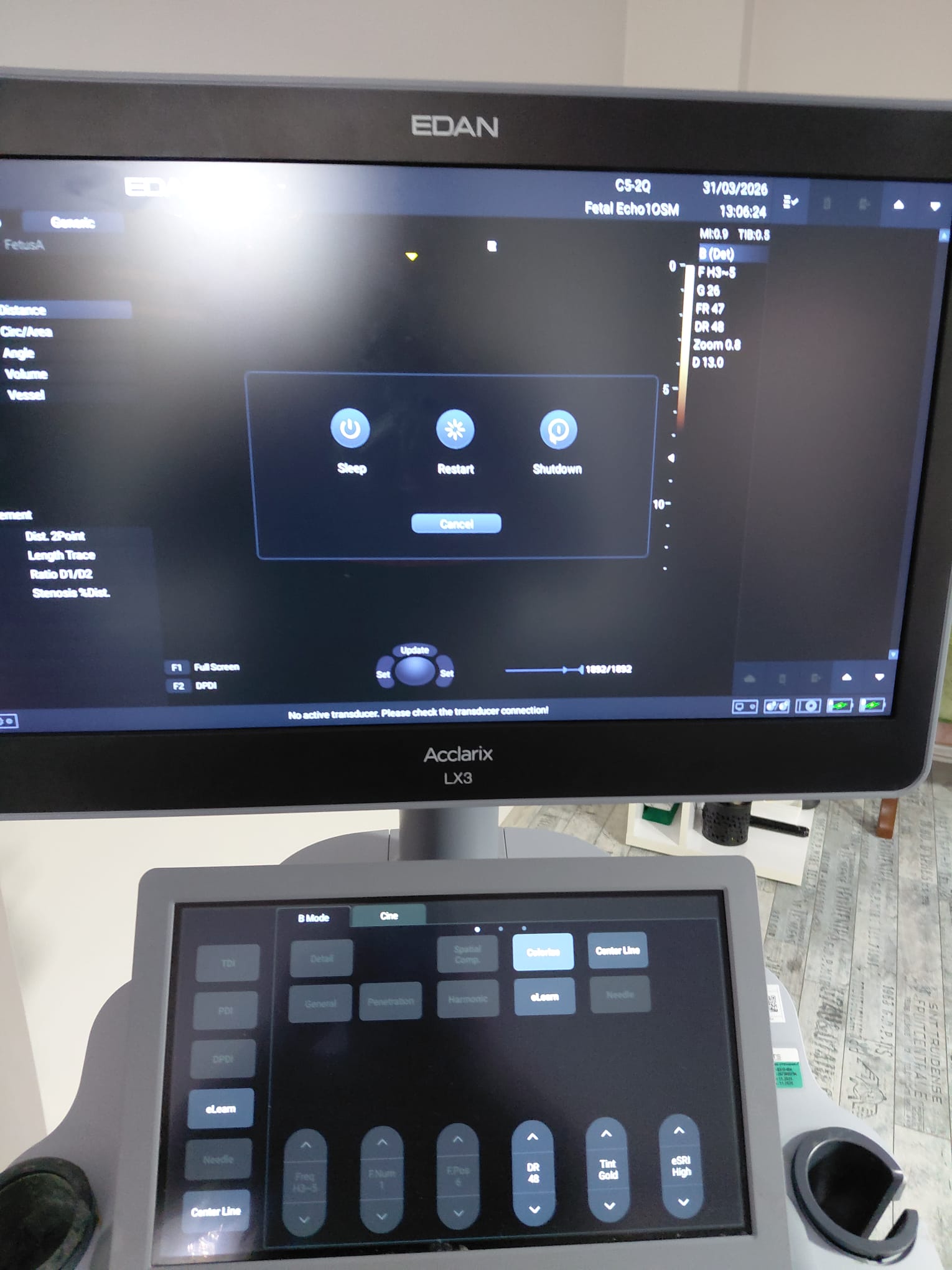
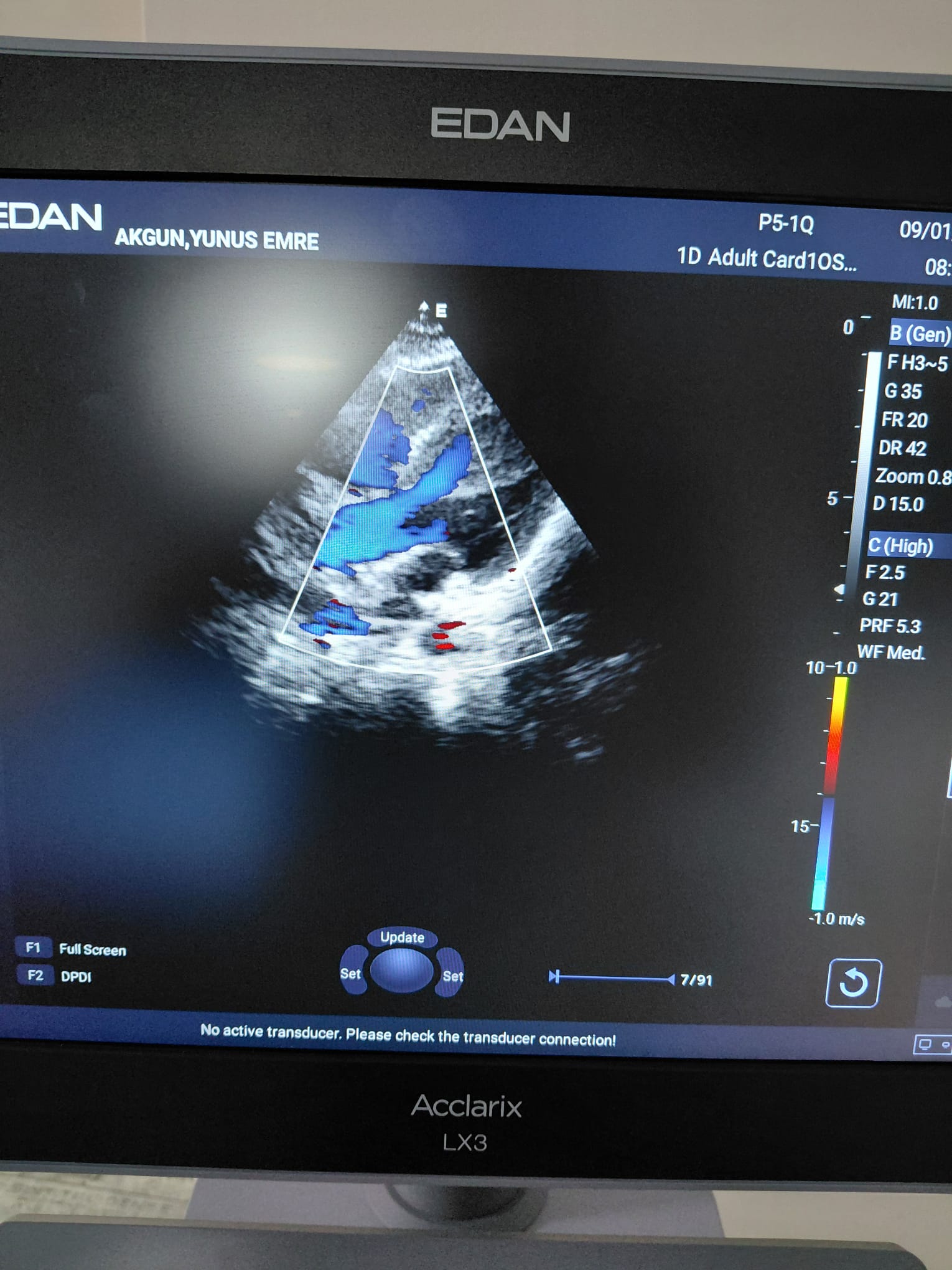
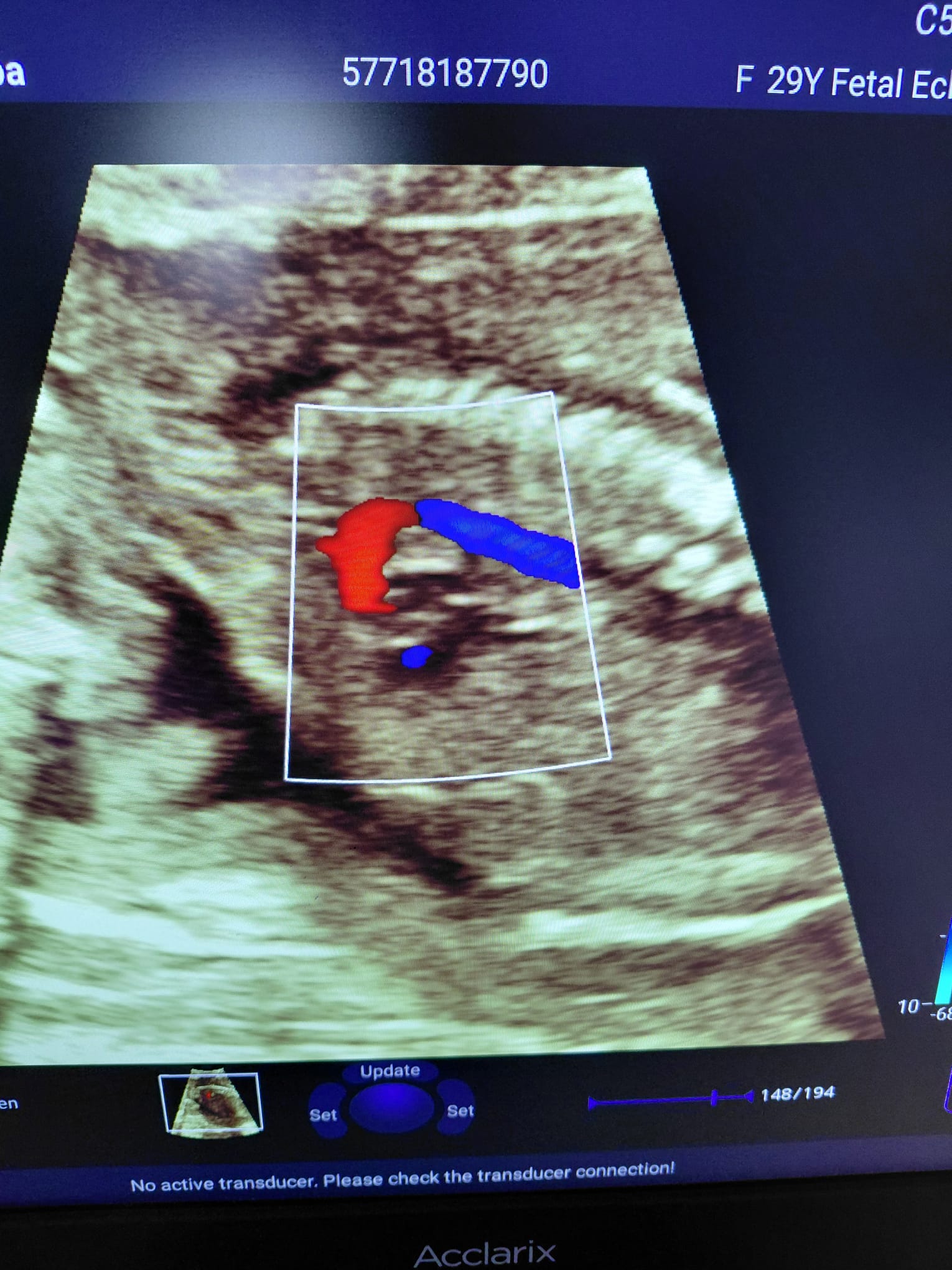
The device’s main monitor and the touch control panel at the bottom are in working condition. No dead pixels or color shifts have been observed on the screens. Color Doppler ultrasound images, patient information, date/time data, and probe frequency information are clearly legible on the main screen. The menu buttons on the touch panel appear active and illuminated.
Existing Faults and Potential Risks
A warning message, No active transducer. Please check the transducer connection! (No active transducer. Please check the transducer connection!), is continuously displayed at the bottom of the device’s active screen images. This situation could be due to the probes not being fully connected to the device during scanning, or it could indicate an electrical communication issue in the device’s probe socket inputs or the probe connectors. Other than this, no physical fault risks have been observed on the device.