Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 10.02.2026
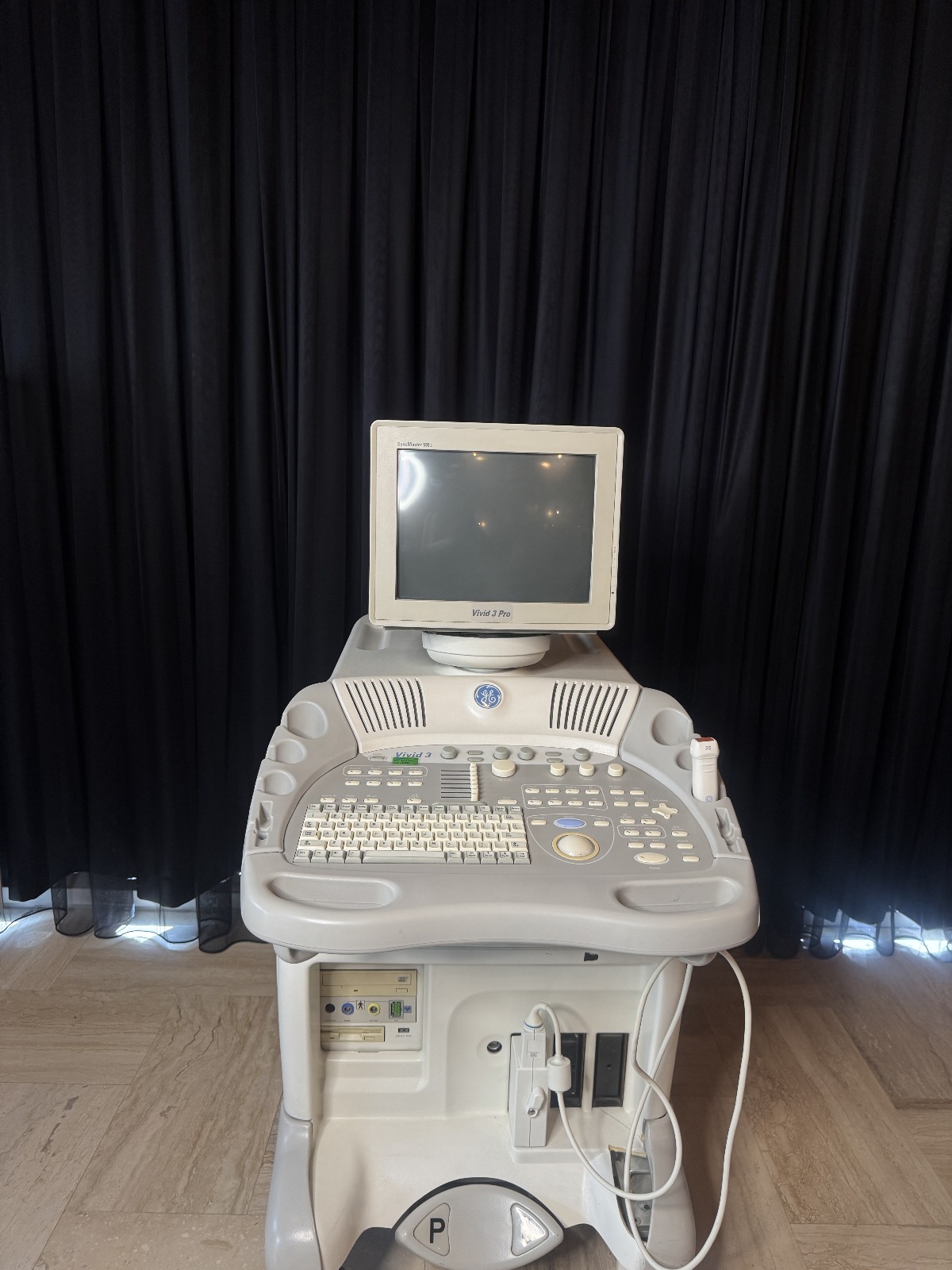
GE Vivid 3 Pro Ultrasound Device Analysis Report
Device Identification
The product examined in the visuals is an ultrasound device, a medical imaging system integrated onto a wheeled stand, featuring a CRT monitor and a manual control panel. The device is an integrated system consisting of a main console, monitor, probe (transducer), keyboard panel, and lower computer/chassis unit.
Brand and Model
Upon detailed examination of the logos and inscriptions on the product, the following brand and model information has been definitively identified:
- Main Device Brand and Model: GE (General Electric) Vivid 3. The inscription “Vivid 3” and the GE logo on a blue background are clearly legible on the upper left part of the console panel.
- Monitor Front Bezel Model: The front bezel of the monitor features the inscription “Vivid 3 Pro”, indicating it is packaged with the ultrasound system.
- Monitor Manufacturer Brand and Model: “SyncMaster 591s” is visible on the upper left corner of the monitor, and the embossed “SAMSUNG” inscription is seen on its back cover. The label information also confirms the Model: 591s.
- Probe (Transducer) Model: The “GE” logo and “3S” model number are clearly legible on the ultrasound probe connected to the system.
Originality
Upon examination of the device’s control panel, logos, labels, monitor compatibility, and the craftsmanship quality of the chassis structure, the product is observed to be an Original GE (General Electric) production. The components and brand labels on it comply with industrial standards.
Areas of Use
The GE Vivid 3 series is an ultrasound system primarily designed for cardiovascular (heart and vessel) imaging. However, considering the 3S probe (Phased Array) it has, it is suitable for use in the following areas:
- Adult Cardiology (Echocardiography)
- Abdominal Imaging
- Transcranial Imaging
It is used for diagnostic purposes in hospitals, cardiology clinics, and imaging centers.
Quantity Information
The visuals show 1 (one) main ultrasound unit and 1 (one) probe (3S) attached to the device.
General Condition
The device is in a used condition. Its second-hand condition is evident from the color changes on the keypads and superficial marks on the chassis. The overall structure of the device is intact; it does not present a disassembled or scattered appearance.
Physical Deformations
- Chassis: While generally appearing solid, there are usage-related friction marks and light dirt, especially on the lower side panels and wheel guards.
- Plastic Components: Yellowing (aging) over time is observed on the white/beige plastic parts of the control panel and on the keyboard keys.
- Cables: The power and data cables at the back of the device are disorganized, but no deep tears or breaks in the cable sheaths have been visually detected.
Mechanical Components
The wheel structure is in place, but there is dirt accumulation on the wheels. The rotary knobs (around the trackball), slider potentiometers (TGC keys), and QWERTY keyboard keys on the control panel appear complete. The Floppy (diskette) drive cover located at the bottom is in place.
Electronic Components
Since the device is in the off position (plugged in but screen off) in the visuals, its electronic functionality could not be tested. The lower module of the device has a diskette drive on the front, and various video outputs and communication ports on the back.
Accessories
- Probe (Transducer): 1 GE brand “3S” model sector (cardiac) probe is available. There is no visible crushing on the probe’s cable, and the connector is attached to the device.
- Cables: A power cable is available.
- Other: No extra printer or recording device (VCR) is present; the corresponding shelf is empty.
Battery Status
This type of stationary ultrasound device operates with mains electricity (220V/110V). No external or visible battery pack has been detected in the visuals. However, the condition of internal components like the BIOS battery cannot be ascertained from the visuals.
Label Information
The data obtained from the labels on the device are as follows:
- Green Label on Control Panel: A green label, assessed to be a calibration or biomedical control label, is present on the device. The dates “26-10-2022” and “10.2023” are legible on the label. This indicates that the device has been under active monitoring until recently.
- Rear Label (Chassis): The large white label features “GE Vingmed Ultrasound”, “Model: Vivid 3”, and electrical warning signs. The serial number is not legible in the visual.
- Rear Label (Monitor): The Samsung label includes safety certifications (UL, CE, CSA) and the model number.
- Probe Label: The inscription “3S” is clear on the probe connector.
Screen Analysis
The device uses an old-type (Cathode Ray Tube) CRT Monitor. The screen is off, so whether there are any error codes, pixel errors, or screen burn marks cannot be determined from the visuals. Although the monitor casing has yellowed, no cracks are visible on the screen glass.
Dimensions and Compatibility
The product is of standard hospital-type mobile wheeled stand dimensions. It has ergonomics suitable for use at the bedside of adult patients.
Year of Manufacture
The exact year of manufacture cannot be read from the label. However, considering the CRT monitor used (Samsung SyncMaster 591s series) and diskette drive technology, it is understood from its technology that the device was manufactured in the early or mid-2000s (estimated between 2002-2006).
Documents
In the visuals, no invoice, user manual, or warranty certificate belonging to the device is present. Only old dated biomedical control labels affixed to the device are available.
Existing Faults
No physically broken, fragmented, or burnt parts are visible in the visuals. There is no warning sign stating “Faulty”.
Potential Fault Risk
Due to the device’s old technology (Diskette drive and CRT screen), the risk of electronic components having reached the end of their lifespan should be considered. The messy and dangling cables at the back may create a risk of tripping or loose sockets during transportation. Yellowing on the keypad may indicate that the device has been exposed to sunlight or heat for a long time, or has been used for a very long period, which could affect key sensitivity.