Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 10.02.2026
GE Vivid 3 Expert Ultrasound Device Analysis Report
Device Identification
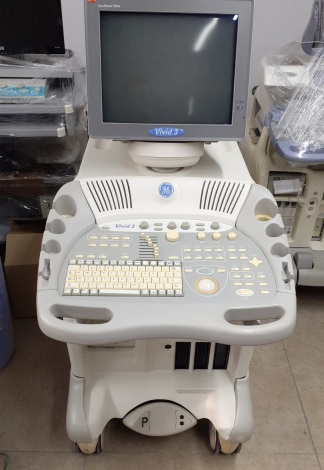
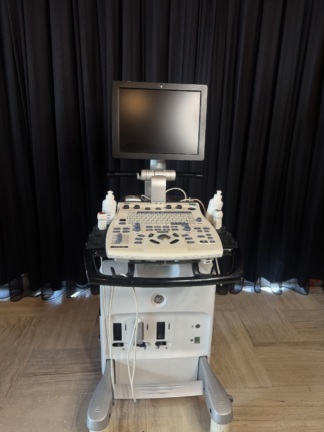
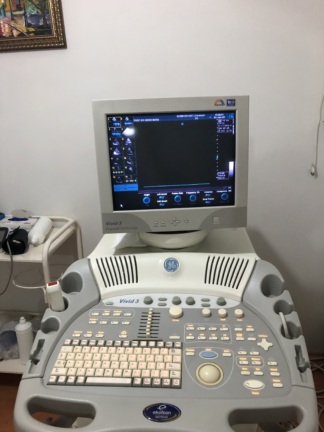
The device in the examined images is a wheeled and mobile ultrasonography system used for medical imaging. The device consists of a main body housing an integrated CRT (cathode ray tube) monitor, a detailed control panel, a processor unit, and data storage drives.
Brand and Model
The data obtained from detailed examinations of the images are as follows:
- Brand: GE (General Electric)
- Model Series: Vivid 3
- Specific Model Designation: Vivid 3 Expert
- Sub-Technology Indication: The phrase “Vingmed Technology” is located on the device’s monitor frame. This indicates that the device incorporates technology from GE Vingmed Ultrasound, specializing particularly in cardiovascular imaging.
- Control Panel: The brand logo on the keyboard is GE (Monogram), and the text “Vivid 3” is also present on the panel.
Authenticity
Upon examining the logos on the product (GE Monogram), model inscriptions (“Vivid 3 Expert”), typical industrial design, color scheme, label placement, and fonts; the device has been identified as an original product manufactured by GE Healthcare. No signs of third-party manufacturing or replica are observed.
Areas of Use
The GE Vivid 3 Expert is a device designed specifically for cardiovascular (heart and vessel) ultrasound applications. The “Vingmed” indication suggests advanced echocardiography capabilities for this device. It is used in hospitals, cardiology clinics, and imaging centers for the following purposes:
- Echocardiography (Heart ultrasound)
- Vascular (Vessel) imaging
- General abdominal ultrasonography
- Small organ imaging
Quantity Information
The images show 1 unit of an integrated ultrasound device.
General Condition
Based on visual inspection, the device is in a used condition. While the product’s overall condition indicates its age, its chassis integrity is preserved. Aesthetically, it shows signs of heavy use.
Physical Deformation and Cosmetic Condition
The following physical conditions were observed on the device’s casing:
- Color Change (Yellowing): Significant yellowing is present on plastic components, particularly on keyboard keys, around the trackball, and on the monitor casing, attributed to age and UV exposure.
- Scratches on Casing: Friction marks are visible on the lower parts where the device’s caster wheels are located, along with dullness on plastic surfaces due to use.
- Monitor Rear: Dust accumulation is noticeable in the ventilation grilles on the rear of the monitor.
- No major cracks, fissures, or serious damage compromising the chassis integrity have been detected.
Mechanical Components
The device has a wheeled structure that provides mobility. The keyboard tray and front handle bar appear solid. The cable management hook and rear handle on the back of the device are in place. The monitor’s swivel mechanism visually appears robust.
Electronic Components and Interfaces
Observations regarding the device’s electronic parts are as follows:
- Front Panel Drives: The module located at knee level on the front of the device contains various media drives. These likely include a CD/DVD drive and slots for older technologies such as MO (Magneto-Optical) disks or floppy disk drives. Additionally, colorful jack inputs resembling microphone/headphone jacks are visible.
- Data Ports: A blue-tipped VGA cable and a grey data cable are seen hanging from the rear of the device.
- Since the device is not displayed in an operational state, no comments can be made regarding its electronic functionality.
Accessories and Missing Parts
- Probe (Ultrasound Transducer): Although probe holders are present on the device, no ultrasound probe is attached to the device or visible next to it in the images. Compatible probes must be acquired separately to use this device.
- Cables: A black power cable is connected to the device. A VGA cable is hanging from the rear.
Battery Status
This type of stationary/console ultrasound device typically operates directly on mains electricity (AC). However, it may contain a small internal battery (CMOS, etc.) for the system clock or BIOS settings. The main power source is the electrical grid; battery operation for mobile use is generally not the primary operating mode for these models.
Label Information
The following information can be read from the label located on the lower rear part of the device:
- Model: VIVID 3
- Manufacturer: GE Vingmed Ultrasound
- Warnings: A yellow-backed safety label containing the phrase “Achtung” (Caution) indicates that the device may have been manufactured for or used in a German-speaking region.
- Specific details such as the serial number and manufacturing date on the label cannot be clearly read due to visual resolution and wear/reflection on the label.
Screen Analysis
The device features a deep-cased, square-shaped CRT (Cathode Ray Tube) monitor, common in the pre-LCD era. The screen is off. CRT screens have a higher probability of experiencing age-related brightness loss or “burn-in” issues compared to LCD screens, but this cannot be determined in the off state.
Size and Compatibility
This is a standard-sized, console-type device designed for adult use, suitable for clinical environments (room-based, not bedside).
Year of Manufacture
Considering the “Vivid 3 Expert” model on the device, the use of a CRT monitor, and hardware features such as floppy/MO drive slots, it is a technical inference that this device was manufactured between the early to mid-2000s (estimated period 2002-2006).
Documents
No user manual, service booklet, invoice, or warranty certificate belonging to the device is present in the images. Due to the device’s age, the factory warranty is no longer valid.
Current Faults
During visual inspection, no distinct cracks or missing parts (excluding probes) that would prevent the device from operating are observed. An “Error” or similar message cannot be displayed as the screen is off.
Potential Fault Risk
- Due to the device being a considerably old model, there is a high probability of age-related fatigue in its electronic components (capacitors, power supply, etc.).
- The trackball (cursor ball) has yellowed; its mechanical sensitivity may have decreased.
- As CRT monitor technology is old, image clarity may fall below current standards, or color fading might occur.
- Without the missing probes, the device cannot perform its imaging function.