Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 04.05.2026
Report code: 1777877059
Edan Acclarix LX3 Diagnostic Ultrasound System Analysis Report
Device Identification
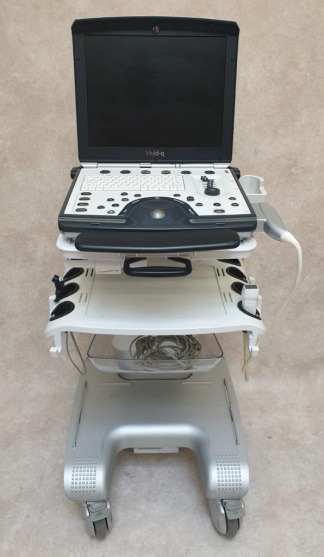
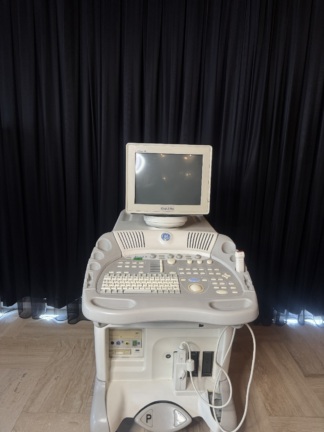
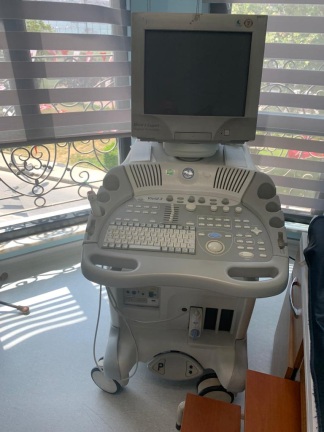
The product in the images is a wheeled, console-type diagnostic ultrasound system used for medical imaging purposes. The device consists of a main unit, a main screen, a touch control panel, and various ultrasound probes.
Brand and Model
The brand of the device is Edan, and the model is Acclarix LX3. This information is clearly legible on the main screen bezel, the lower part of the touch panel, and the rear label. Additionally, this model information is included in the user manual.
Authenticity
The quality of the logos on the device, the layout of the labels, the CE 0123 certification mark, the touch screen interface, and the general workmanship details indicate that the product is authentic.
Areas of Use
This device is used for non-invasive imaging of internal organs and tissues. As understood from the screen captures and probe types, it is suitable for use in the following areas:
- Obstetrics and Gynecology: Fetal imaging (Fetal Echo), obstetric measurements.
- Cardiology: Adult cardiac imaging (Adult Card).
- General Radiology: Abdominal and other soft tissue examinations.
Quantity Information
The images show 1 ultrasound main unit and 3 different types of ultrasound probes. There are a total of 4 pieces of main hardware.
General Condition
The general condition of the device appears to be very good and clean. The screens are active and functional. No significant wear is observed on the plastic components.
Physical Deformation
No scratches, cracks, fractures, or discoloration have been detected on the device casing, main screen, or touch panel. The outer surfaces and cables of the probes also appear clean and intact.
Mechanical Components
The wheels, probe holders, and moving parts of the device appear to be sturdy based on the images. No mechanical issues are visible.
Electronic Components
The main screen and touch control panel of the device are active. Imaging functions are operational. The buttons and parameter setting areas on the touch panel are clearly visible.
Accessories
The following accessories are visible with the device:
- 1 C5-2Q coded convex probe.
- 1 P5-1Q coded phased array probe.
- 1 P7-3Q coded phased array probe.
- 1 Turkish user manual (Diagnostic Ultrasound System User Manual, Version 1.3).
The connector parts of the probes are not visible in detail in the images. There are probe socket inputs available on the device.
Battery Status
There is no visual information regarding whether the device has an internal battery. The rear label only contains AC power input information.
Label Information
The label on the back of the device contains the following information:
- Model: Acclarix LX3
- Serial Number (SN): 560522-M23816170001
- Manufacturing Date: 2023-08-25
- Power Information: 100V-240V~, 50Hz/60Hz, 2.5A-1.2A
- Maximum Mass: 55kg
- Certifications: CE 0123, MD (Medical Device)
- UDI Codes: (01)06944413813298, (11)230825, (21)560522-M23816170001
Screen Analysis
The main screen displays images and menus for different imaging modes:
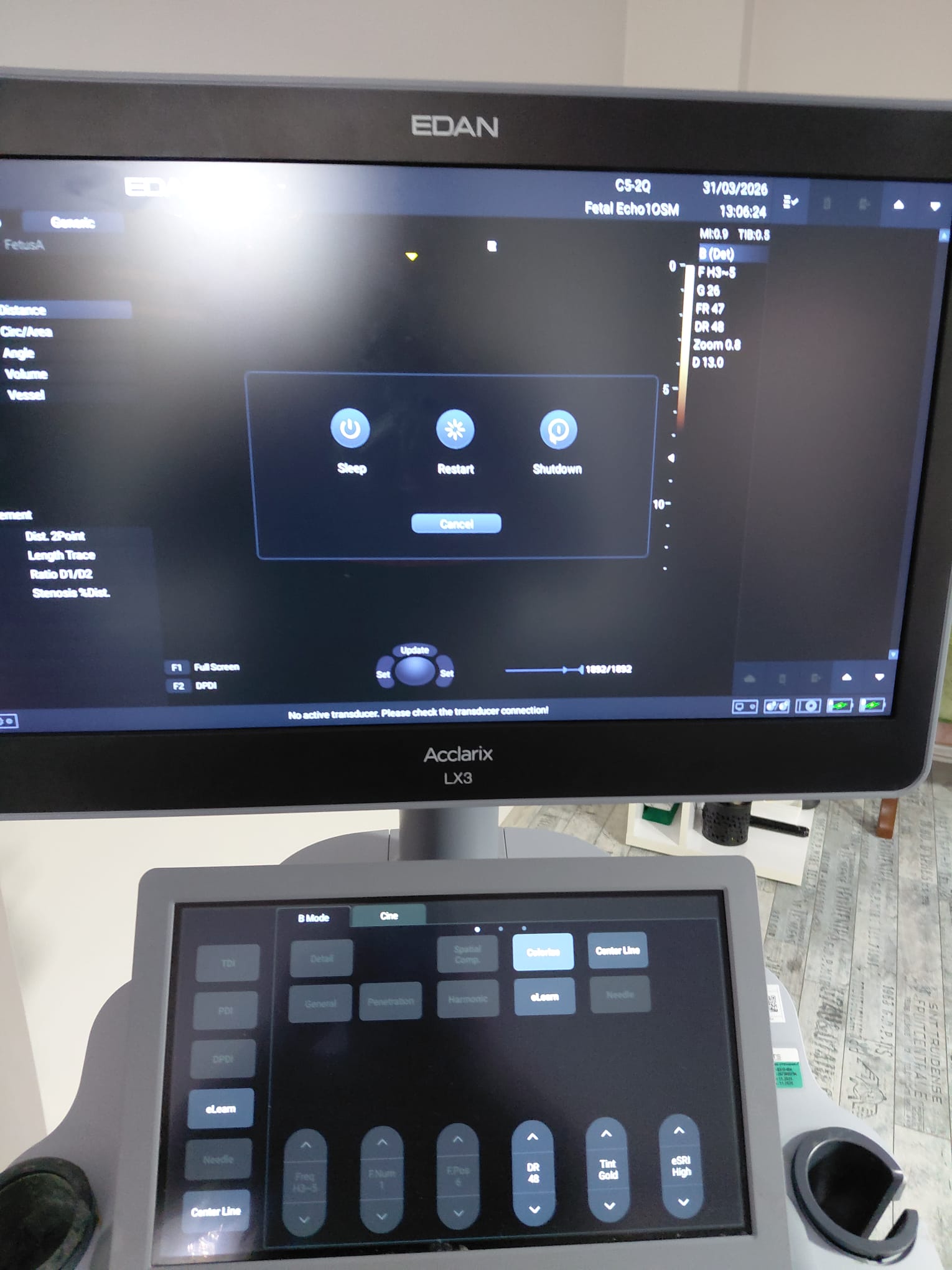
- In one image, a power menu containing “Sleep”, “Restart”, and “Shutdown” options is open. At the bottom, the warning “No active transducer. Please check the transducer connection!” is visible.
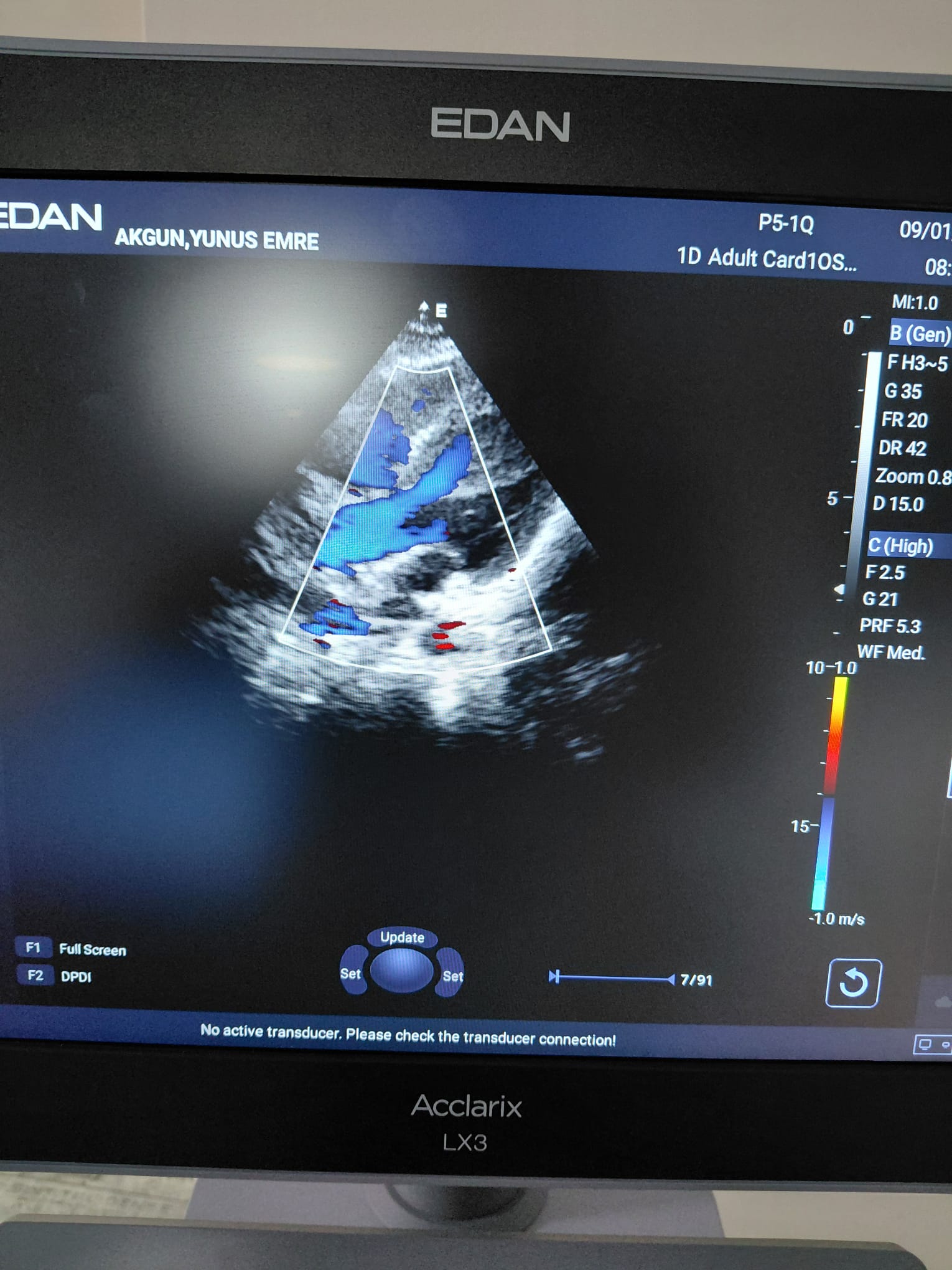
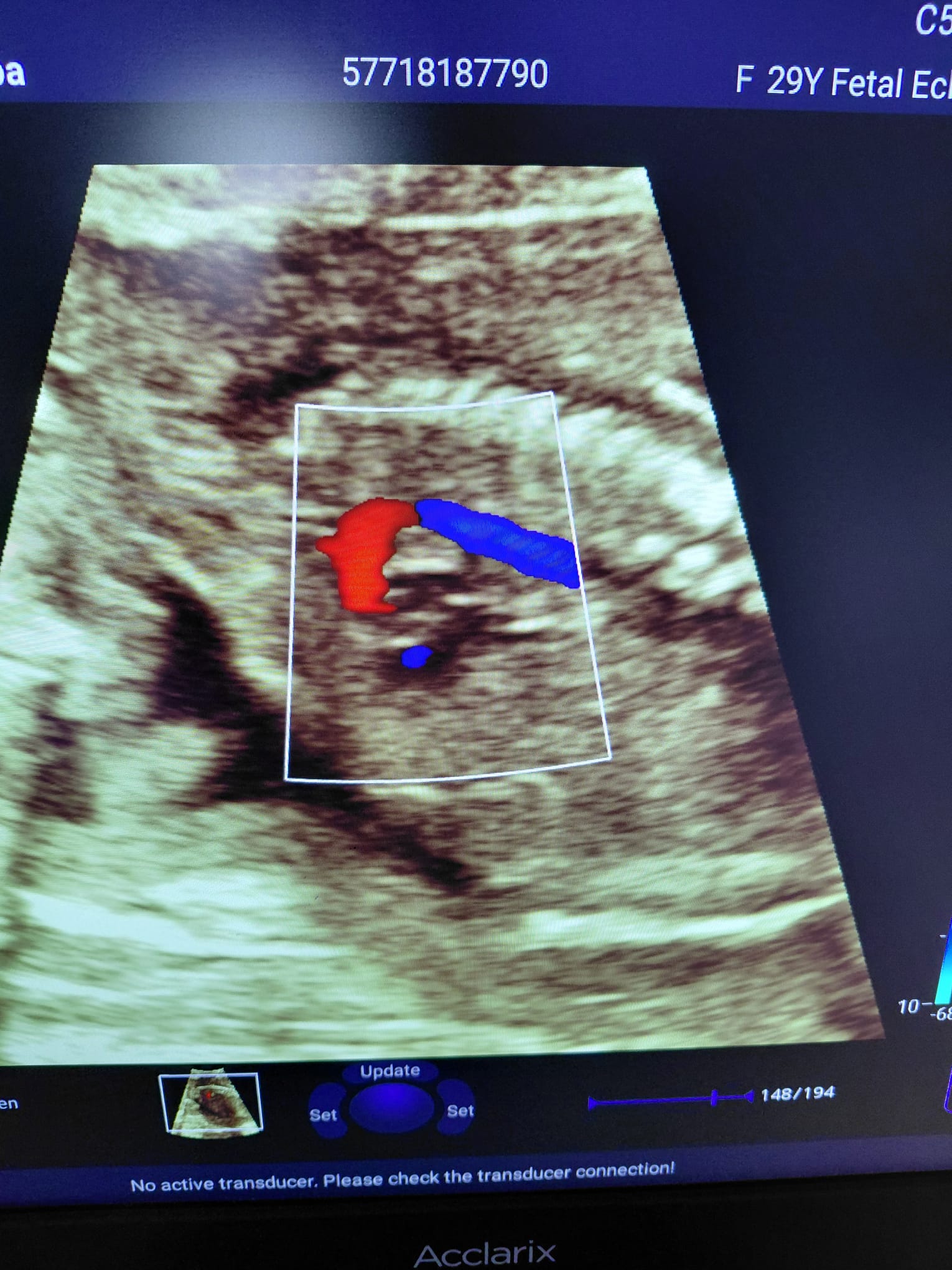
- Other images include color Doppler images related to cardiac and fetal examinations, patient name (AKGUN, YUNUS EMRE), examination dates (31/03/2026, 09/01/2026), and technical parameters (MI, B Mode, G, FR, DR, Zoom, etc.).
Dimensions and Compatibility
The device’s probes are compatible with adult (Adult Card) and fetal imaging. The device itself is of standard console-type ultrasound dimensions.
Manufacturing Year
Next to the manufacturing symbol on the rear label of the device, there is a date of 2023-08-25. The device was manufactured in 2023.
Documents
The Turkish user manual for the device is visible in the images. No invoice or warranty certificate is present in the images.
Usage Duration
There is no information on the screen or label regarding the total operating hours of the device.
Existing Faults
There is no clear evidence of a fault, such as broken parts or error messages, in the images. The “No active transducer” warning seen on the screen is a normal situation resulting from no probe being selected or connected at that moment.
Potential Risk of Failure
No negative conditions such as severe wear, corrosion, or crushed cables were observed in the images. The condition of the device appears good, which indicates that the potential risk of failure is low.