Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 07.09.2025
GE Vivid 7 Dimension Ultrasound Device Analysis Report
Overview and Product Description
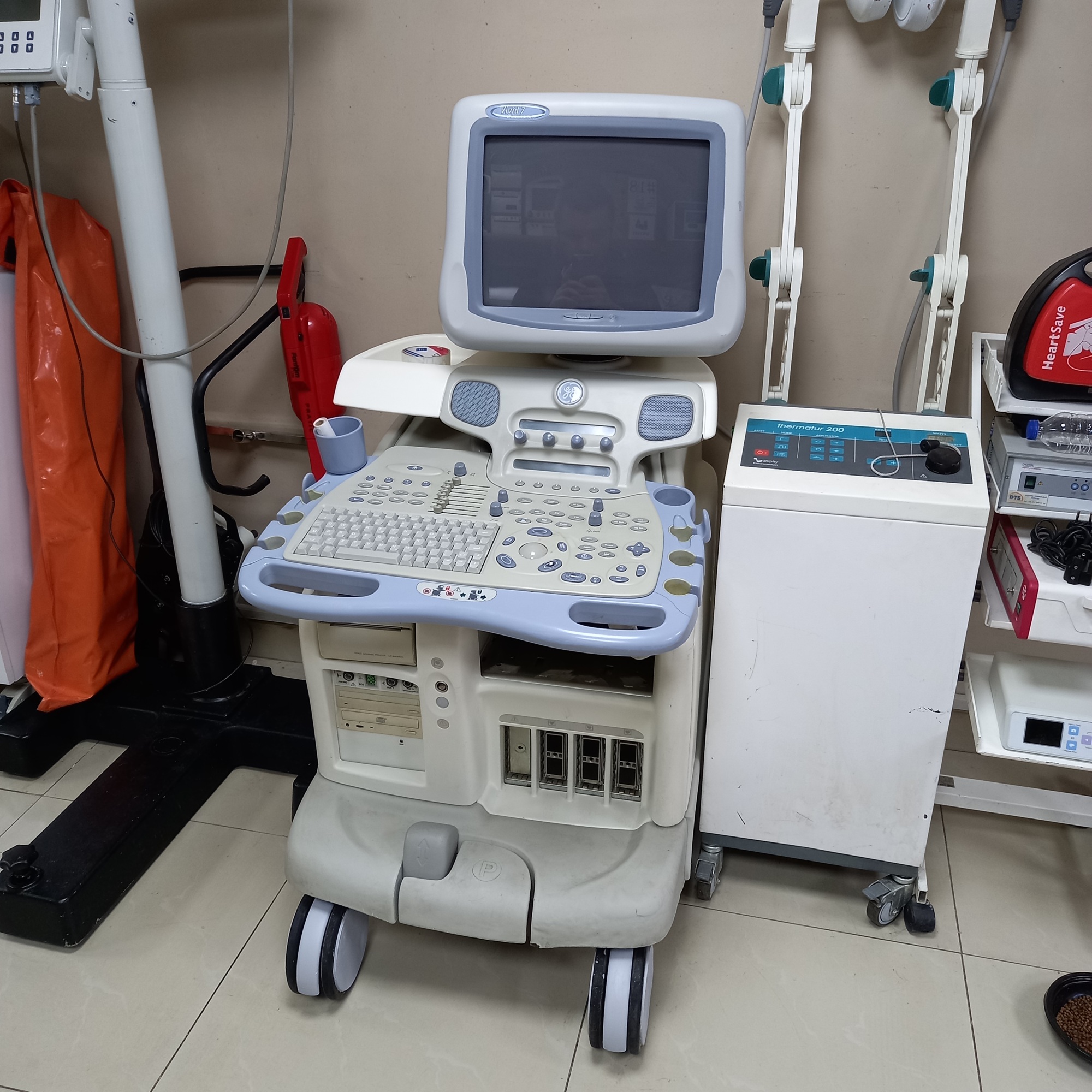
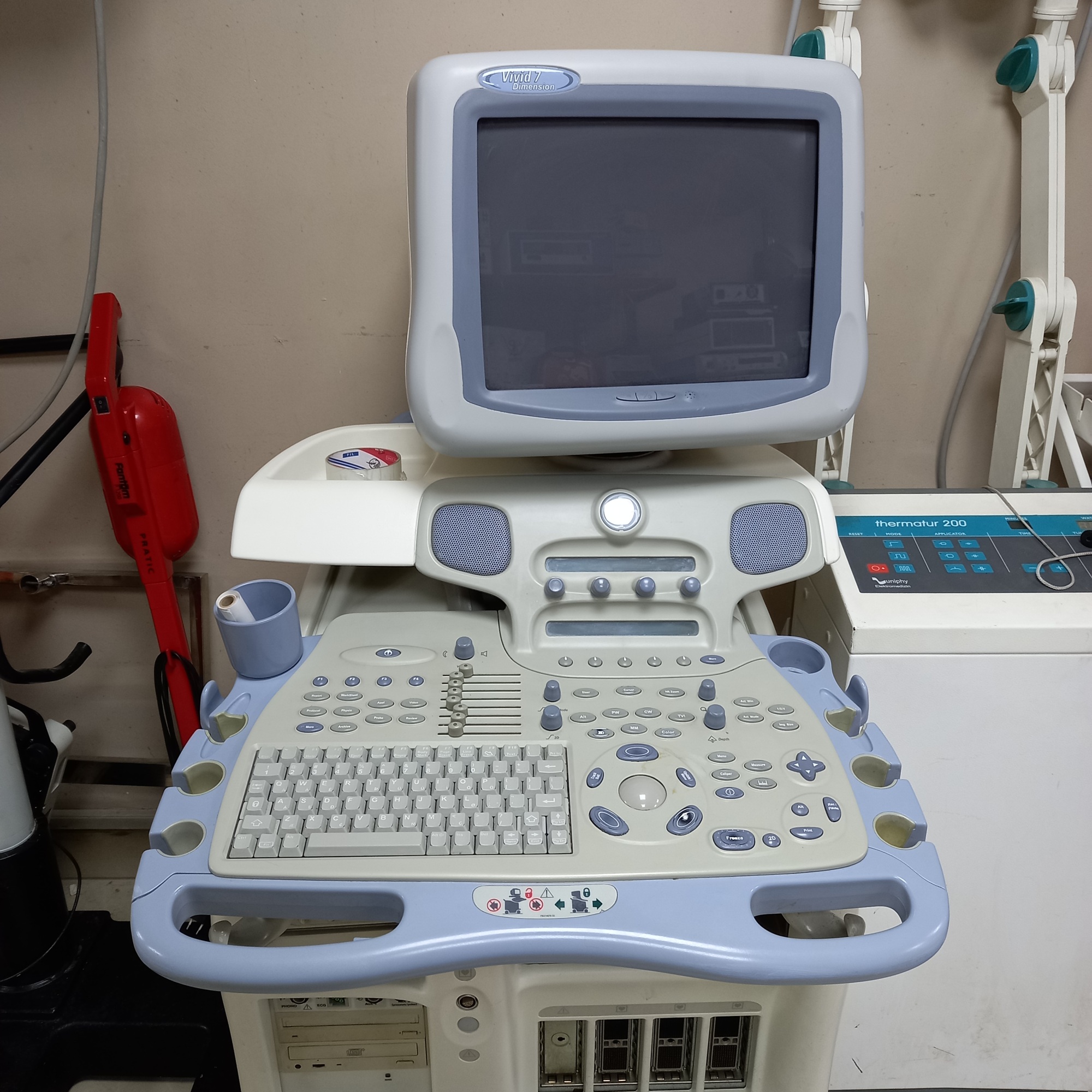
The product shown in the visuals is a complete ultrasonography device integrated onto a wheeled stand. The device consists of a main console, user interface (keyboard and control buttons), monitor, and an ultrasound probe. It is a system used for medical diagnosis, enabling the imaging of internal organs. From the brand and model information, it is understood that it is specifically designed for cardiovascular imaging. The general structure of the device indicates its suitability for professional clinical use.
Brand, Model, and Authenticity Determination
As a result of detailed examinations, the brand and model of the device have been definitively identified. The text “Vivid 7 Dimension” is clearly visible on the monitor frame of the device. The GE (General Electric) logo is located on the side of the casing. Based on this information, the brand of the product is GE, and the model is Vivid 7 Dimension. The integrated video printer’s brand is Sony, and its model is UP-895MD. The quality of the logos and general craftsmanship details indicate that the product is original.
Physical Condition and Deformation Analysis
The overall condition of the device is assessed as used. Various physical deformations have been observed due to use.
- Casing: The beige plastic casing of the device shows yellowing and color variations in places. Especially in the lower front section, above the wheel, there are paint chips and noticeable scratches.
- Control Panel: A visible crack and a broken piece of plastic are present in the ultrasound probe housing on the control panel. Slight dirt accumulation is observed around the keypad.
- General Cleanliness: It is understood that the device is generally dusty and requires thorough cleaning.
Mechanical and Electronic Component Examination
Upon visual inspection of the device’s mechanical and electronic components, the following observations were made:
- Mechanical Components: The device has four wheels, which appear physically sound. The buttons, switches, and trackball on the control panel are in place.
- Electronic Components: The device’s monitor is off in all visuals, therefore, whether it is working or if there are any issues with the screen cannot be visually confirmed. There is one CD/DVD drive and a Sony brand video printer on the main casing. Four probe connection ports are visible on the device, but only one probe is attached in the visuals.
Accessories and Connection Ports
In the visuals, a total of one product and one accessory are present with the device.
- Product Count: 1 GE Vivid 7 Dimension ultrasound device.
- Accessory Count: 1 ultrasound probe (with the inscription “3S”).
An integrated Sony UP-895MD Video Graphic Printer is located at the bottom of the device. Various input/output ports such as PHONO, AUDIO (L/R), S-VHS, VIDEO, and AUX 1-2 are present on the rear panel. No external cables or other accessories are included in the visuals.
Labels and Technical Information
The labels on the device have been examined in detail, but information such as serial number, lot number, or REF code could not be clearly identified. The only readable model information belongs to the integrated printer, Sony UP-895MD, and the 3S code on the probe.
Summary and Conclusion
This report is based on a visual analysis of a GE Vivid 7 Dimension ultrasound device. The device is used and has cosmetic flaws such as scratches, paint chips, and yellowing on its casing. Additionally, a break in the probe housing has been identified. Since the device’s screen is off, no comment can be made regarding the functionality of its electronic components. One 3S model ultrasound probe and an integrated Sony printer are visible with the product. In its current state, the device is considered a product that needs to be inspected and tested on-site by potential buyers. Aside from the apparent physical defects, no serious potential malfunction risk was visually detected.