Ready For Sale
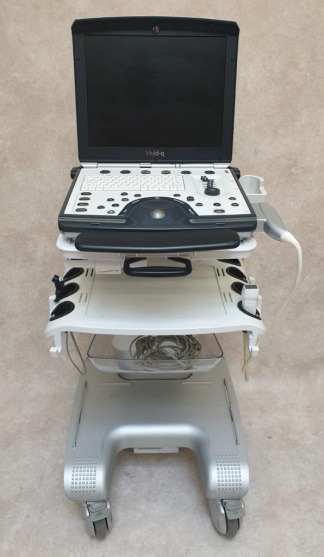
Secondhand GE Vivid S5 Echocardiography Machine
Price: USD$ 16.500,00 Approx: 742.500,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 10.02.2026
GE Vivid S5 Ultrasound Device Analysis Report
Device Identification
The product analyzed in the images is a professional ultrasonography device mounted on a mobile stand (cart), used for medical diagnosis and imaging. The device consists of an integrated monitor, control panel (console), processor unit, and a wheeled stand structure providing mobility.
Brand and Model
As a result of detailed examination of the images, the following brand and model information has been definitively identified:
- Brand: GE (General Electric) – The iconic “GE” monogram logo is clearly visible on the lower body of the device.
- Model: Vivid S5 – The model name “Vivid S5” is clearly legible and written in the lower right corner of the control panel. Additionally, design lines belonging to the GE Vivid series are present in the brand label area on the side of the device.
Originality
Upon examining the GE logo on the product, the button layout on the control panel, material quality, the design language specific to the Vivid series, and the font characteristic of the “Vivid S5” inscription, the product is evaluated as an original GE (General Electric) production. No signs of a counterfeit or replica product (such as a faulty logo, poor quality plastic joints, etc.) have been observed.
Areas of Use
This device (GE Vivid S5) is primarily a system specialized in cardiovascular (heart and vessel) imaging. However, it can also be used in general-purpose ultrasonography applications. Its areas of use include:
- Cardiology (Adult and Pediatric Echocardiography)
- Vascular Imaging
- Abdominal Imaging
- Obstetrics and Gynecology (OB/GYN) and Urology examinations
Quantity Information
The inventory shown in the images is as follows:
- 1 unit of GE Vivid S5 Ultrasound Main Console and Stand
- 1 unit of LCD Monitor (Integrated into the device)
- 1 unit of Ultrasound Probe (Held in hand and seen in the probe holder in the image, likely a 3S-RS sector probe)
- 2 units of Ultrasound Gel Bottles (In the holders at the back of the device)
General Condition
The device is cosmetically used (second-hand). However, its general integrity has been preserved. No breakage, missing parts, or severe damage is immediately apparent. It is understood from the signs of use that it has been actively used in a hospital or clinic environment.
Physical Deformations
During the visual inspection, the following points are noteworthy:
- The plastic components of the device show slight discoloration (yellowing) that can occur over time and superficial use dirt.
- Slight wear due to use can be observed on the wrist rest (palm rest) areas of the control panel, but no deep cracks or breaks have been detected.
- No significant erasure is visible on the keypad and the texts around it.
Mechanical Components
Analysis of mechanical components:
- Monitor Arm: The articulated arm to which the monitor is attached appears sturdy and maintains the screen’s position.
- Wheels and Stand: The base structure of the stand appears sturdy (although the wheels are not visible in detail, the base structure maintains its integrity).
- Gel and Probe Holders: The gel bottle storage areas at the back of the device and the probe holders on the sides are intact.
Electronic Components and Control Panel
Electronic condition analysis based on the images:
- Screen: The device’s LCD monitor is physically sound; no deep scratches or cracks are visible on the screen surface. Since the screen is off in the images, no comment can be made regarding pixel defects or image quality.
- Keypad and Trackball: The QWERTY keyboard is complete. The TGC (Time Gain Compensation) sliding potentiometers (sliders on the bottom right) are in place and their covers are present. The trackball is seated in its housing. Rotary knobs appear complete.
Accessories and Connection Points
Accessories and inputs on the device:
- Probes: Upon examining the probe attached to the device and held in hand, it appears to be a sector (phased array) probe, likely used for cardiac imaging. The inscription “3S” is legible on the probe’s connector socket (usually indicating a 3S-RS model). The cable sheath appears intact, and no crushing or breakage is discernible in the image.
- Ultrasound Gel: There are 2 ready-to-use ultrasound gel bottles on the device.
- Socket Inputs: Empty slots and socket input areas, believed to be for peripheral units (printer, recorder, etc.), are visible on the front lower panel of the device. It is not clear whether there are missing covers or accessories in these areas, but the slots are empty.
Label Information
The rear label containing the device’s serial number is not clearly shared in the images. However, the technical data obtained from the image is as follows:
- Model: Vivid S5
- Probe Code: The inscription “3S” on the probe connector (3S Sector Probe)
- Panel Buttons: Standard GE Vivid interface function buttons such as Patient, Probe, Review, Report, Protocol are present.
Battery Status
Vivid S5 models generally operate with AC mains electricity, but some versions may include batteries to keep the system in sleep mode during transport. As no power cable supplying energy to the device or active battery indicator is visible in the images, there is no visual data regarding the battery’s presence or health.
Potential Failure Risk
Based on the images, no critical damage or potential failure risk (such as cable peeling, broken screen, rusted connectors, etc.) has been identified. The device can be evaluated as being in good condition according to second-hand medical product standards. However, since the piezoelectric crystal structure of probes and cable conductivity in medical devices cannot be fully determined visually, technical service inspection is recommended.