Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 09.02.2026
Deymed Diagnostic TruTrace 4 EMG System Analysis Report
Device Identification
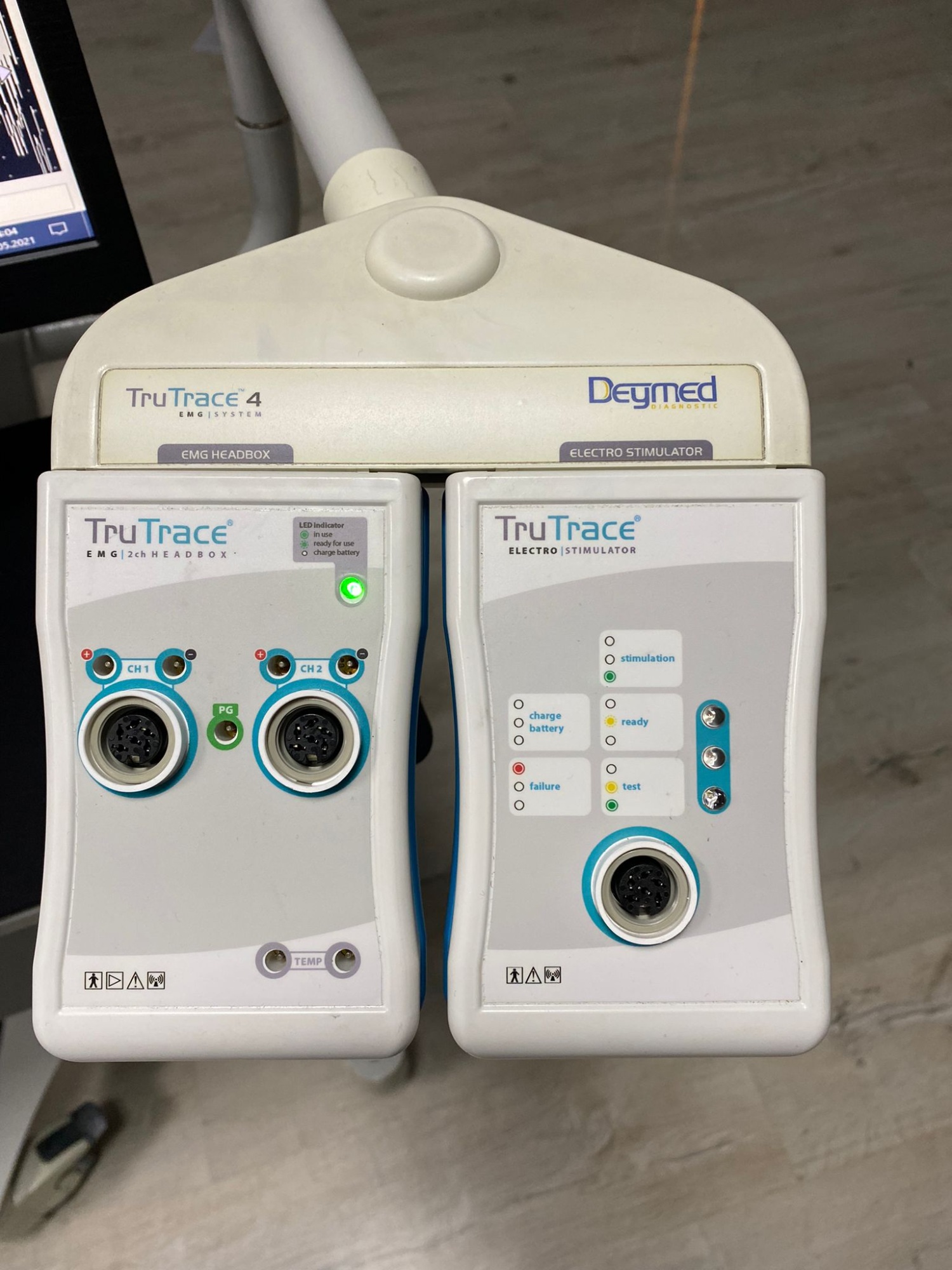
The system shown in the visuals is a professional medical diagnostic device mounted on a wheeled mobile station, used in neurological and neuromuscular diagnoses. The system consists of a desktop computer, monitor, input devices (keyboard/mouse), a mobile transport cart (trolley), and specialized modules for collecting signals from/providing stimuli to the patient. The product has been identified as an Electromyography (EMG) and Nerve Conduction Study (NCS) system.
Brand and Model
As a result of detailed analyses of the visuals, the brand and model information of the device, with 100% certainty, is as follows:
- Main System Brand: Deymed Diagnostic
- Main System Model: TruTrace 4 EMG System
- Module 1 (Amplifier): TruTrace EMG 2ch HEADBOX (2-channel EMG headbox)
- Module 2 (Stimulator): TruTrace ELECTRO STIMULATOR (Electro Stimulator)
- Monitor Brand: Philips
Originality
Upon examining the logos on the device (Deymed Diagnostic, TruTrace), the quality of typography, the molding craftsmanship of the modules, and the label placements, the product appears to be entirely original. The Philips monitor and Deymed components are original productions compliant with industry standards.
Areas of Use
This system is used in Neurology and Physical Therapy departments for the following purposes:
- EMG (Electromyography): Measurement and recording of electrical activity of muscles.
- NCS (Nerve Conduction Studies): Measurement of the speed and ability of peripheral nerves to transmit electrical signals.
- EP (Evoked Potentials): Evoked potential tests.
The device is used in clinical settings for the diagnosis of conditions such as neuropathies, muscle diseases, and nerve compressions.
Quantity Information
The visuals show 1 fully equipped station. The components of the station are as follows:
- 1 Deymed module holder arm and bracket system
- 2 Pluggable Modules (1 Headbox, 1 Stimulator)
- 1 Philips Monitor
- 1 Transport Cart (Trolley)
- 1 Desktop Computer Case (on the lower shelf of the trolley)
- 1 Keyboard and 2 Mice (1 standard, 1 ergonomic round/medical mouse could be)
General Condition
The device is shown installed and in working condition. Its general condition is “Used – Good”. It gives the impression of being actively used in a clinical environment. There are no signs of heavy dirt or rust, and it appears well-maintained.
Physical Deformation
During the inspection, no cracks, fissures, deep scratches, or significant discoloration (yellowing) were detected on the device casing, monitor, or white plastic module casings. No bending is observed in the mechanical arm from which the modules are suspended.
Mechanical Components
The device’s wheeled transport cart (trolley) appears sturdy. The connection points of the monitor and module holder arms seem rigid. The modules are properly seated in the bracket. The table surface and keyboard area appear mechanically stable.
Electronic Components
The visuals prove that the system is electronically active and operational:
- Monitor: The screen is on, displaying a Windows-based EMG software interface. No pixel errors or screen cracks are visible.
- Modules: The green LED indicators on both modules (Headbox and Stimulator) are lit. The “in use” light on the left module and the “ready” light on the right module are active, indicating that the device is powered and ready for communication.
- Computer: It is clear that the case on the lower shelf is providing display to the system.
Accessories
Accessories included in the system or visible in the image:
- White wired keyboard and white wired mouse.
- A large black mousepad in front of the screen.
- On the lower leg of the cart, there is an accessory bag that appears unopened. The “Deymed” brand (readable as Degmed in reverse) and “Reusable Grounding Strap” text can be discerned on it.
- Power adapters and multi-socket power strips are present under the trolley.
Battery Status
On the “TruTrace ELECTRO STIMULATOR” module on the right side, there is a “charge battery” indicator and a battery icon. This indicates that the stimulator unit has an internal battery. The green light being on suggests that the battery is full or that the device is operating without issues while powered by mains electricity.
Label Information
Technical label data read on the modules are as follows:
- Top Console: “TruTrace 4 EMG | SYSTEM”, “EMG HEADBOX”, “ELECTRO STIMULATOR”
- Left Module: “TruTrace EMG”, “2ch HEADBOX”, Input ports: CH1, CH2, TEMP. Safety Class Symbol: BF (Body Floating).
- Right Module: “TruTrace ELECTRO | STIMULATOR”, Indicators: Stimulation, Charge Battery, Ready, Failure, Test. Safety Class Symbol: BF.
Screen Analysis
The image on the Philips monitor resembles a real-time patient recording session or a demo mode:
- Software: Deymed EMG interface.
- Menu Items: Anatomical regions such as “Forearm”, “Elbow”, “Upper arm”, “Axilla” are listed.
- Graphic: A yellow-colored graph of intense noise or muscle activity (MUAP or interference pattern) is flowing in the lower right part of the screen.
- Date/Time: The date “26.05.2021” and time “04:04” are read on the Windows taskbar.
Size and Compatibility
The device is dimensioned for adult use, but it may also have the software infrastructure for pediatric measurements depending on the probes used (as far as can be understood from the screen, the current setting is for adult anatomy).
Year of Manufacture
The year of manufacture is not visible on the physical labels. However, the system date of 2021 on the screen indicates that the device was active on this date. The product is likely a model manufactured between 2018-2021.
Period of Use
A hardware counter is not present in the visuals. The date on the software showing the year 2021 suggests that the device has been in use since at least that date or that the system clock has not been updated.
Potential Malfunction Risk
The only potential risk factor observed in the device is cable management. The somewhat loose and dangling cables (adapters and connection cables) on the cart could pose a risk of getting caught in the wheels or stressing the sockets during transport. Other than cable organization, no serious risk, breakage, or oxidation has been observed.